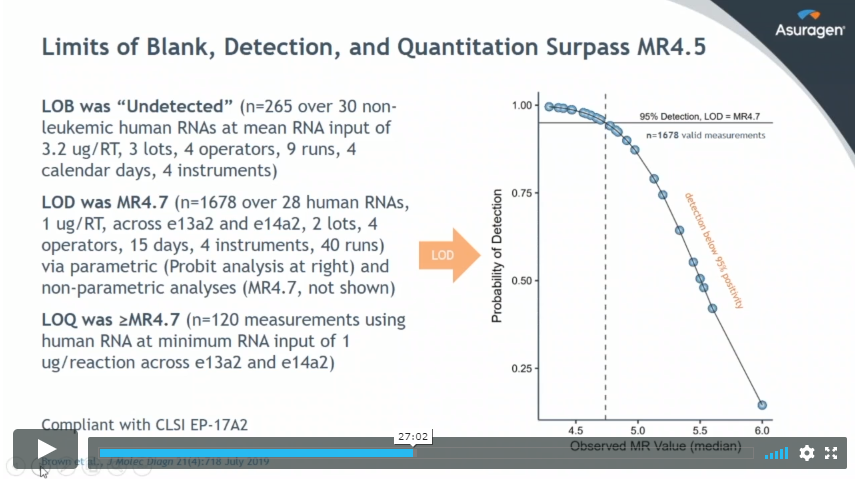
To mark the launch of the ground-breaking Quantidex® QPCR BCR-ABL IS Kit, manufacturer Asuragen held a series of three webinars. We covered the first webinar here, so check that out first if you haven’t already seen it. The second webinar, below, examines the clinical expectations for BCR-ABL monitoring against the backdrop of the launch of this new product.
Webinar series
The second webinar is delivered by Justin Brown, PhD, Principal Scientist at Asuragen. It focuses on:
- Review of the clinical expectations for BCR-ABL monitoring
- Overview of the QuantideX® qPCR BCR-ABL IS Kit
- Discussion of key analytical validation studies and performance data and how the assay aligns to clinical needs
To view the webinar, please click the below image:
Or this link: https://www2.asuragen.com/l/223762/2019-11-07/hzzl2
QuantideX BCR-ABL IS Kit provides labs with a robust and reliable method for monitoring leukaemia patients, also allowing them to keep pace with the advances in TKI therapy.
It is seen as a major breakthrough for labs dealing with the monitoring of leukaemia patients, offering reduced complexity and optimised workflow without losing high-quality performance.
You can read about the QuantideX® qPCR BCR-ABL Portfolio in more detail here – but we would strongly recommend listening to/watching the fascinating webinars, too, which are delivered by some of the world’s leading figures in oncology and haematology research and product development.
For more information about the QuantideX® qPCR BCR-ABL Portfolio, contact VH Bio today.
Enquire about this article
"*" indicates required fields