
WILDLIFE: Highlights from the VH Bio 2026 Company Retreat
The VH Bio team recently gathered for our annual company meeting to reflect on the last year of trading, plan for the year ahead, and celebrate our team spirit through team building [...]

BSHI/EFI 2026: A Belated Scottish Welcome!
BSHI came a little earlier than usual this year with it being held jointly with the EFI conference. Although it was kept local with it being held in an unusually sunny, spring-time blossom-filled [...]
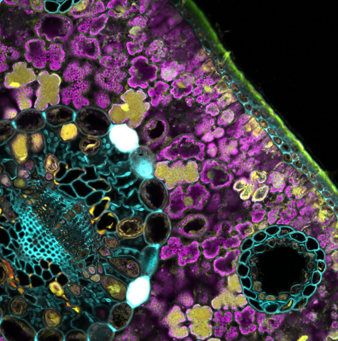
OptiSlides Webinar: Colour, Clarity & Confidence for Microscopy
High-quality, prepared microscope slides from Luxidea, now available in six channel colours! Is your microscope showing you the full picture? Meet VH Bio and Professor Kamala Patel, CEO of Luxidea, in this webinar [...]

The Epitope-Analysis Toolbox: Selecting the Right Tool for the Job
From Antigens to Epitopes: Refining HLA-Antibody Risk Assessment The wide adoption of virtual crossmatching to assess donor/recipient immunological risk has necessitated a focus on HLA-antibody definition beyond the traditional antigenic-level. Increased access to allelic-level donor and recipient HLA-typing, in combination with tools enabling greater definition of HLA-antibody profiles, can aid in more nuanced [...]
H5N1 Avian Influenza: Ensuring Sample Integrity and Biosafety in Emerging Zoonotic Threats
Intro Highly pathogenic avian influenza A (H5N1) continues to require attention from UK diagnostic and surveillance laboratories. While sustained human-to-human transmission has not been observed, the virus remains present in wild birds [...]

High Sensitivity, Faster Results: Clinical Validation of the geneLEAD VIII Automated RT-PCR System
Real-World Performance Backed by Peer-Reviewed Data Clinical laboratories and research institutions are under increasing pressure to deliver molecular diagnostic results quickly and accurately. The geneLEAD VIII automated sample-to-result RT-PCR platform addresses this need, combining automation with robust analytical performance [...]

Protocol for Calm:
an activity book for scientists who think too muchWe're delighted to share our new colouring in and activity book: Protocol for Calm. Designed for reflection, not perfection, this book offers up a selection of colouring in patterns and brain teasers [...]

How to Study the Brain Using Cell Lines
Why Use Cell Lines to Study the Brain? The brain is a highly complex organ and can develop equally complex diseases. A robust, cost-effective, and accessible way to investigate biological processes and [...]

Microchimerism Matters: Advancing HSCT Monitoring Through NGS
Overview Allogeneic haematopoietic stem cell transplantation (HSCT) remains the only curative therapeutic option for many haematological malignancies, including acute myeloid leukaemia (AML), acute lymphoblastic leukaemia (ALL), and myelodysplastic syndromes (MDS). Post-transplant monitoring [...]

The Future of Metagenomics in Clinical Diagnostics
Highlights from the IBMS Congress 2025 on advancements in metagenomics in microbiology As the year draws to a close, the IBMS Congress in Birmingham offered an ideal moment to reflect on [...]

Minority Report: Why detecting microchimerism can make the biggest difference
In the evolving field of transplantation and histocompatibility testing, chimerism monitoring remains a fundamental part of post-transplant care. However, the tools we use to measure donor and recipient DNA are evolving. The [...]

Emerging Markers for Precision Monitoring & Personalised Care in Transplantation
Current challenges in transplantation In organ donation and transplantation, our primary concern is to treat the patient whose organ is failing and, if doing so with a transplant, avoid acute rejection. For [...]
NewsAlison Wortley2025-04-15T10:47:42+00:00