
Brief intro to culture for the ID of pathogens in routine molecular pathogen diagnosis
The standard method used in clinical microbiology laboratories for detection of bacterial and fungal pathogens over the last century is culturing of clinical specimens obtained from the patients. The supply of nutrients promotes the growth of potentially present microorganisms which is detected by automated signalling systems in response, for instance, to certain CO2 levels produced during growth. Gram staining gives a first direction to the selection of more specific antibiotics. Ultimately, biochemical and microbiological fingerprinting analysis leads to the identification of species followed by antibiotic susceptibility testing.
What are the limitations of culturing for pathogen diagnosis?
In some infectious episodes, the causative microorganism cannot be detected by culturing due to the relatively low sensitivity of the technique, even though the clinical signs and symptoms are suggestive of an infection. The occurrence of false culture-negative results, when microorganisms are in fact present, can be due to prior antibiotic treatment, slow growing or fastidious microorganisms, or the presence of viable but nonculturable bacteria. Culture-negative results are for instance seen with infective endocarditis, bone and joint infections including foreign body associated infections, otitis media, and meningitis.
The whole process is time consuming, taking on average 2 to 7 days, depending on the organism, before results are obtained, and up to 50% of infections are missed by blood culture while 15-25% of results are negative. In cases of suspected sepsis and bloodstream infection, each hour of delayed treatment increases mortality risks by nearly 8%.
These limitations show how routinely used blood culture methods are not an ideal gold-standard, as results often come too late, are incomplete or not sensitive enough and can be misleading and relatively labour-intensive. There is a crucial unmet need to shorten as well as improve current laboratory procedures for the detection and identification of microorganisms. In recent times, molecular-based technologies have emerged that address the limitations of culture methods and offer effective diagnosis.
Automation in routine molecular pathogen diagnosis is crucial for the provision of timely and accurate results. Handling errors with resulting false findings can be greatly reduced by these new technologies.
Is your work in the lab affected by the limitations of culture for microbe detection?
One such solution for culture-independent identification of bacteria and fungi is realised with Molzym’s molecular diagnostic technology, MicroDx. Designed to rapidly detect over 1,300 bacteria and fungi, Molzym’s diagnostic technology delivers added value in both diagnostic sensitivity and time.
Instead of culturing and cell proliferation, Molzym’s automated molecular pathogen diagnosis technology selectively isolates microbial DNA and broad-range PCR or Real-Time PCR amplifies 16S/18S gene targets in bacteria and fungi for detection and downstream sequencing analysis and identification. Molzym’s molecular diagnostic technology can identify pathogens days earlier than culture and in patients who are negative with culture (Kühn et al. 2011; Orszag et al. 2013). In comparison with similar technologies, Molzym’s molecular diagnostic technology can remove DNA from host cells and dead microbes, as well as limiting contaminated reagents, columns, and polymerases to provide better accuracy and more sensitive results.
Molzym’s technology allows to diagnose true infections by identifying pathogens in culture-negative patients, with an increase in positivity rate by between 40-67%, and with the capacity to identify up to two times more pathogens than culture.
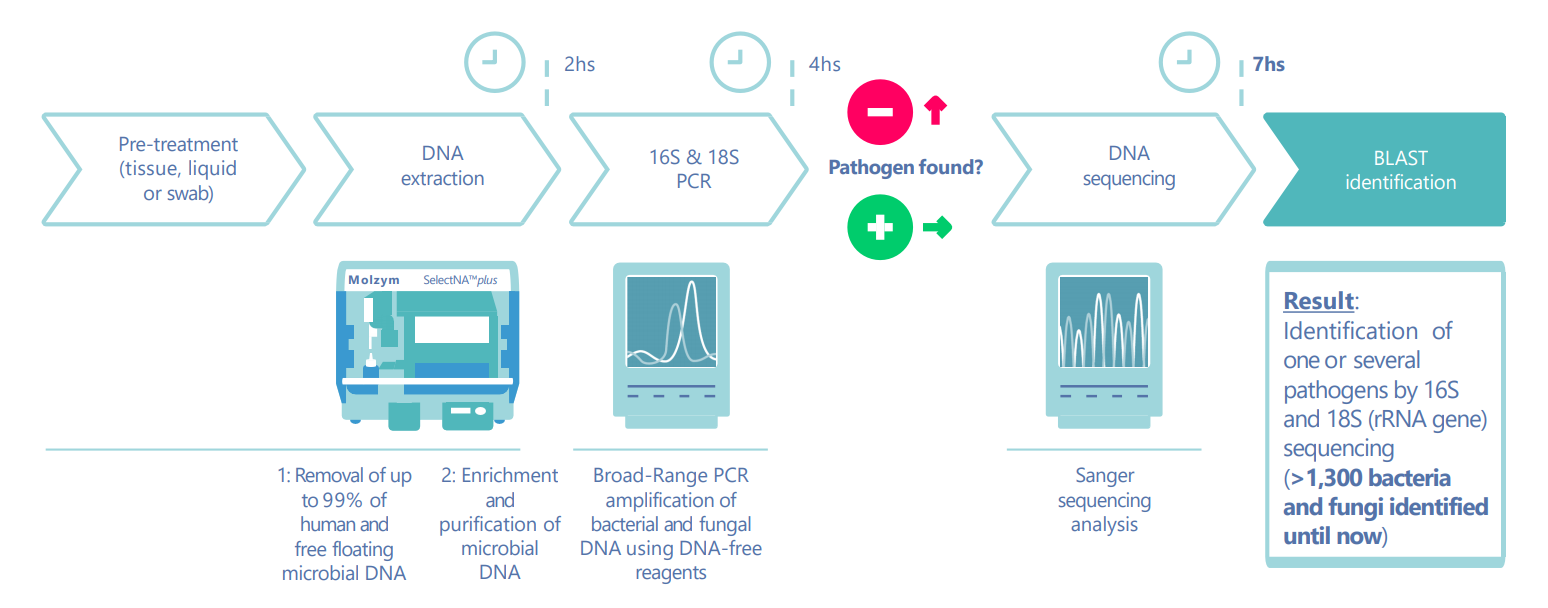
The clinical diagnostic workflow is shown in fig.1:
Fig. 1: After 4 hours, which includes manual or automated microbial DNA extraction and broad-range 16S/18S PCR amplification, initial results indicate if bacteria or fungi are present in the sample. Positive amplicons are applied to Sanger sequencing and the pathogens are identified by using Molzym’s online tool SepsiTest™-BLAST which can complete the analysis within approximately 7 hours.
About the technology
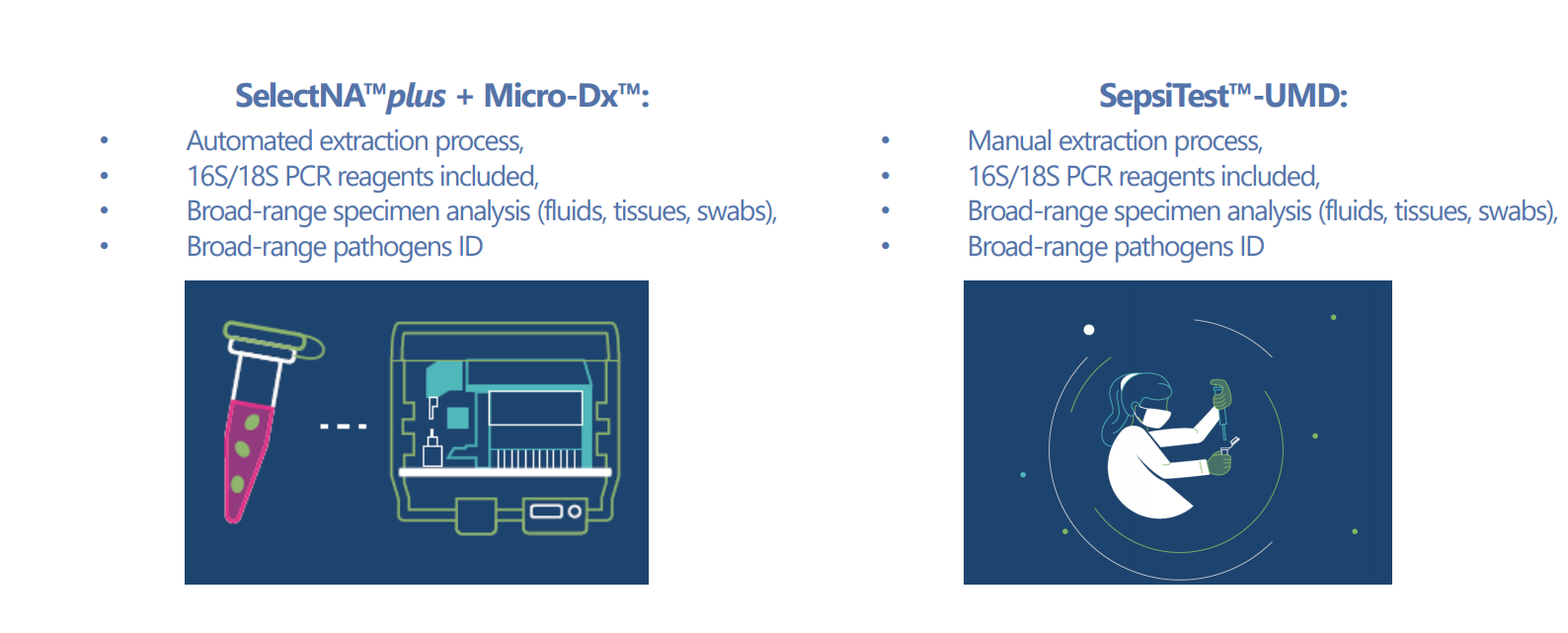
VH Bio supplies Micro-Dx™ from Molzym, which offers unique automated pathogen DNA extraction and broad-range molecular analysis of bacteria and fungi. The technology delivers fast and precise culture-independent diagnosis by innovative pathogen enrichment and broad range PCR and sequencing. Micro-Dx™ provides low hands-on, real-time PCR diagnosis in just one protocol for a diversity of clinical specimen.
SepsiTest™-UMD follows, on a molecular level, the line of the microbiological pathway from sample to result. Unlike culture which needs days to results, SepsiTest™-UMD can identify pathogens within hours, including those prevailing but not proliferating in the body due to antibiotic inhibition or not growing because of fastidious growth demands.
The SelectNA™plus instrument is a fully automated, walk-away pathogen ID solution which is run with the Micro-Dx™ kit containing DNA-free reagents and consumables for the simultaneous extraction of microbial DNA from up to 12 samples. The kit further contains DNA-free reagents for the broad-range amplification of parts of the 16S and 18S rRNA genes of eubacteria and fungi, respectively.
Simple, Comprehensive Pathogen ID Solutions
Discover the diagnostic workflow for infectious diseases in 60 seconds:
WEBINAR: Management of bacterial and fungal infections: Efficiency of culture-independent molecular testing
This webinar presented by Molzym’s COO Dr. Michael Lustig explores the limitations of conventional cultures and the culture-independent approaches available within direct molecular testing. Watch the webinar to learn about the diagnostic value and clinical impact of broad-range 16S & 18S rRNA analyses.
About Molzym
Molzym’s fast and accurate clinical diagnostic solutions are culture-independent, free of reagent-borne contaminations and applicable for the identification of pathogens from a great variety of specimens in just 7 hours.
About VH Bio
VH Bio Limited is a leading supply and distribution company for the life sciences industry within the United Kingdom and Republic of Ireland, specialising in key areas of science including Transplant Diagnostics, Molecular Biology, Immunology and Cell Biology.
Contact VH Bio to learn more about Molzym’s product range.
References:
Enquire about this article
"*" indicates required fields