Copy number variations in SMN1 and SMN2 are, respectively, associated with the onset and severity of spinal muscular atrophy (SMA), a debilitating and life-threatening illness of the central nervous system. Recent studies have demonstrated that transmission risk and disease severity may be impacted by the presence of additional variants, such as SMN1 gene duplication events and disease modifier in SMN2.
The AmplideX® PCR/CE SMN1/2 Plus Kit* revolutionizes the analysis of these two genes by delivering comprehensive results in less than four hours. Powered by AmplideX technology, the assay accurately quantifies SMN1 and SMN2 exon 7 copy number and also detects SMN1 gene duplication and SMN2 disease modifier variants – all from a single reaction. The assay shares a common workflow with other assays in the AmplideX product portfolio and is optimized for use on widely established laboratory equipment.
Features & Benefits
Reduced Complexity
- Similar workflow to AmplideX PCR/CE FMR1*† kit eases implementation and training
- Multiplexed, scalable design allows analysis of single-nucleotide variants, small indels, and copy-number changes from a single PCR reaction
- Assay-specific software automates variant calls and streamlines data analysis
Optimized Workflow
- DNA-to-data in less than four hours with only 60 minutes of hands-on-time
- Optimized for use on commonly installed CE equipment
- Fully-kitted solution sourced from a single vendor
Quality Results
- Ability to differentiate between 0, 1, 2, 3 and ≥4 copies for both SMN1 and SMN2
- Automated variant and copy-number genotyping
- Accuracy demonstrated through comparisons with multiple orthogonal methods
*For Research Use Only. Not for use in Diagnostic procedures.
†CE-IVD for US Export Only.
Analytical Characteristics
- Automated reporting of SMN1/2 copy number and variant detection via AmplideX Reporter Software
Figure 1. Example Electropherogram Output – AmplideX PCR/CE SMN1/2 Plus Kit

- DNA to data in less than four hours – all from a single PCR reaction
Figure 2. AmplideX PCR/CE SMN1/2 Plus Kit Testing Workflow

- Excellent concordance of SMN1 and SMN2 copy number results to sites’ in-house methods for over 500 replicates
Figure 3. Reproducibility of SMN1 and SMN2 copy number reporting across sites for over 500 replicates.
- High assay specificity permits detection of SMN1–SMN2 hybrid peaks, including those resulting from gene conversion events
Figure 4. Detection and differentiation of SMN1 and SMN2 hybrid peaks.
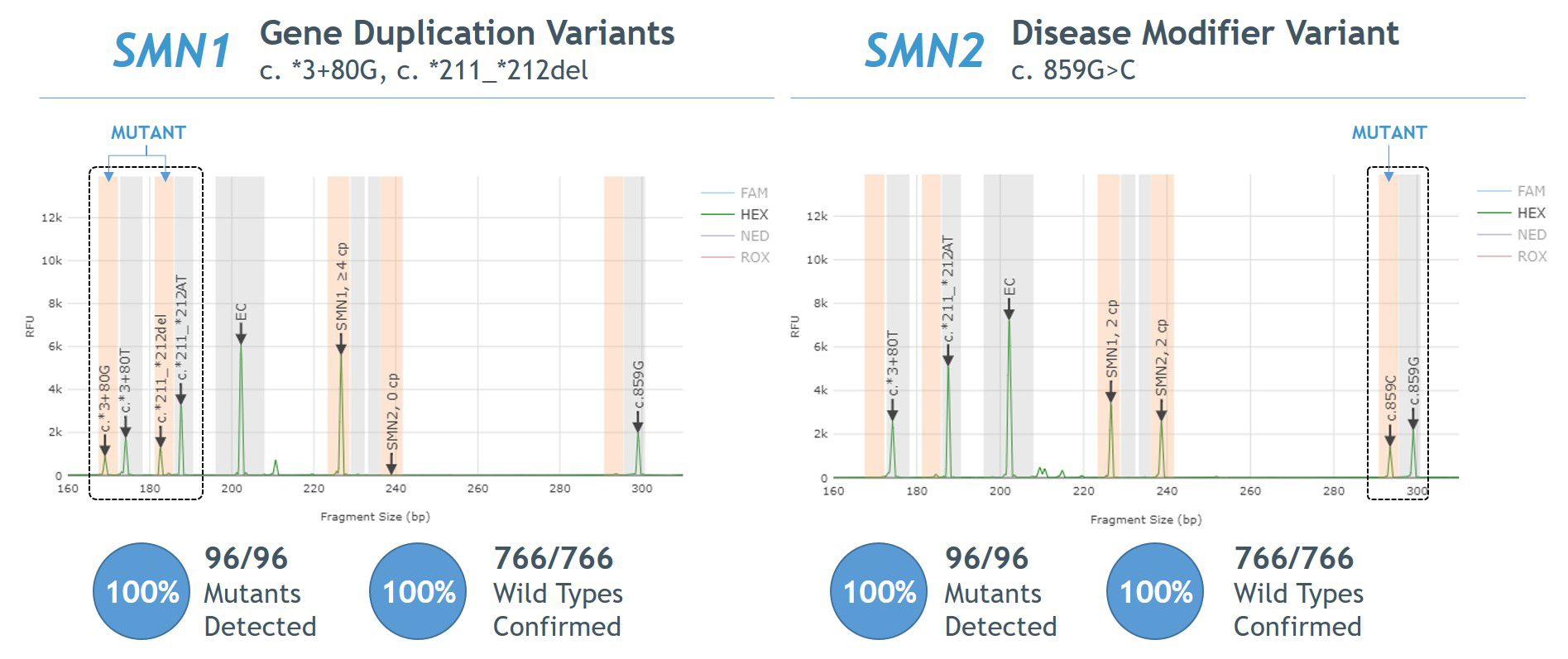
- 100% agreement between Sanger sequencing and PCR/CE SMN1/2 Plus Kit for detection of SMN1 gene duplication and SMN2 disease modifier variants
Figure 5. Agreement between AmplideX PCR/CE SMN1/2 Plus Kit and Sanger sequencing for SMN1 gene duplication and SMN2 disease modifier variants
Posters
A Rapid Diagnostic and Screening System for Spinal Muscular Atrophy that Reports Copy Number Changes, Single Nucleotide Variants and Small Indels
AmplideX® SMA Plus Kit
SKU CODE: 06A00050-VIRTUAL
| PRODUCT | Units |
|---|---|
| AmplideX® SMA Plus Kit06A00055 | 50 Reactions |
| AmplideX® SMA Plus Kit06A00056 | 100 Reactions |
Showing 2 Products
Showing 2 Products
Enquire about this product
"*" indicates required fields

