The success and effectiveness of Tyrosine Kinase Inhibitors (TKIs) for treatment of Chronic Myeloid Leukaemia (CML) has altered clinical expectations as patients live longer, healthier lives.
This success has, in itself, created new challenges, however: clinical practice guidelines now encourage the use of highly-sensitive molecular assays in order to track CML patient progress on TKI therapy. Lengthened lifespans mean patients are now more likely to be monitored in different labs over the course of their disease, creating potential consistency issues.
This has created an acute need for a sensitive, precise and reliable testing solution. Bare minimum expectations/requirements of a BCR-ABL assay now include:
- Detection of deep molecular responses and earlier signs of relapse, at least MR4.5 (0.0032%IS) in RNA from clinical specimens
- Providing robust and reliable results, demonstrating reproducibility of results (low %CV) across broad quantitative range
- Report results on the IS and calculate ABL1 copy number, enabling accurate reporting on the IS and confirming assay sensitivity with copy number verification
Introducing the QuantideX® qPCR BCR-ABL Portfolio
This is where the QuantideX® qPCR BCR-ABL Portfolio from Asuragen comes in. It’s the best-in-class solution for assessment of major and minor BCR-ABL fusion transcripts when monitoring CML patients.
QuantideX® qPCR BCR-ABL delivers:
- Common workflows that streamline testing and implementation
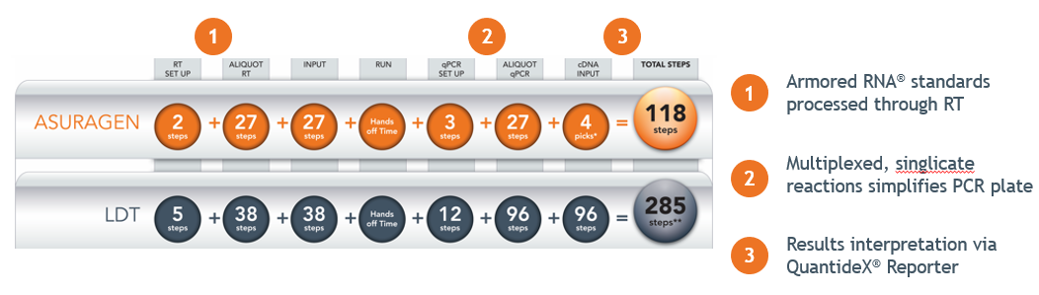
- Multiplex designs that simplify plate setup and maximise throughput [fig. 1]
- Analytical validation with clinical specimens ensures clinical relevance
- Complete, end-to-end solutions from sample to result
With a streamlined workflow with limited hands-on time, the QuantideX® qPCR BCR-ABL offers improve operating efficiency with reduced risk of operator error. [Fig. 2]
Other benefits include reduced complexity with software that provides automated results reporting, with no calculations required, while the IS Kit features direct reporting on the International Scale (IS) – eliminating the need for sample exchange – as well as ABL copy number reporting.
An optimised workflow means fusion and control genes are amplified and detected in the same reaction and the ‘singlicate’ testing format facilitates maximum throughput scalability.
But this would be worthless if it wasn’t for the high-quality results: the QuantideX® qPCR BCR-ABL range has an ultra-sensitive limit of detection (MR4.7 and LR4.61 for the IS and minor kits, respectively), determined using human RNA from clinical specimens, not cell lines [Fig. 3].
Cell lines express higher levels of ABL1 than human RNA: both the IS and minor kits showed considerable increases in sensitivity in cell lines compared to clinical samples. Using cell lines may overestimate the LOD, increasing the risk of false negatives.
The armoured RNA®-based standards provide true RNA quantification, achieved as a result of the standards/calibrators going through the same RT and qPCR reactions as the patient samples.
For more information about the QuantideX® qPCR BCR-ABL Portfolio, contact VH Bio today.
Enquire about this article
"*" indicates required fields