Asuragen recently showcased their latest product developments via a live corporate satellite and scientific poster presentation at the European Human Genetics Virtual Conference ESHG 2020.2, which ran from June 6-9. The videos are now available for on-demand viewing, below.
The ESHG annual event moved to a virtual platform in response to the COVID-19 pandemic and featured numerous scientific presentations. In their session, Asuragen demonstrated how their growing family of AmplideX® testing kits can provide simple and streamlined solutions for the analysis of complex genomic targets.
Using AmplideX® Technology to Detect Single Nucleotide Polymorphisms, Indels, and Copy Number Variations
Asuragen’s corporate satellite, “Looking Beyond Repeat Expansions: Using AmplideX® Technology to Detect Single Nucleotide Polymorphisms, Indels, and Copy Number Variations,” showcased the new AmplideX® SMA Plus Kit via a live Satellite Session and Scientific Poster. Both the Satellite and Poster reinforced the high performance and versatility of Asuragen’s foundational AmplideX® chemistry in detecting a broad spectrum of variants and variant types – all while preserving the same, simple, single-day workflow you have come to appreciate.
During the satellite session, Henny Lemmink, PhD, Clinical Laboratory Geneticist, Department of Genetics, UMC Groningen, described how testing for spinal muscular atrophy spans both diagnostic and screening settings. Recent studies have demonstrated that SMN1 and SMN2 copy number information alone may not be sufficient to guide appropriate action for either testing scenario.
In the diagnostic setting, reporting on the unique c.859G>C SMN2 disease modifier variant may help to refine disease prognosis. Additionally, identifying “silent carriers” is of increasing demand when screening for asymptomatic carriers.
Lemmink discussed his lab’s experience using the AmplideX SMA Plus Kit. The kit reports SMN1 and SMN2 copy number as well as SMN1 gene duplication and SMN2 disease modifier variants to inform on silent carrier status and disease prognosis, respectively, in less than four hours.
Watch the presentation: Rapid, Comprehensive, and Complete SMN1/2 Profiling with the AmplideX® SMA Plus Kit (CE-IVD)
https://www2.asuragen.com/l/223762/2020-06-29/mtnzb
John Milligan, Senior Scientist at Asuragen, also conducted a video presentation of one of the company’s scientific posters at the meeting, which further highlighted how the innovative assay is highly informative for patients for diagnosis and carrier screening:
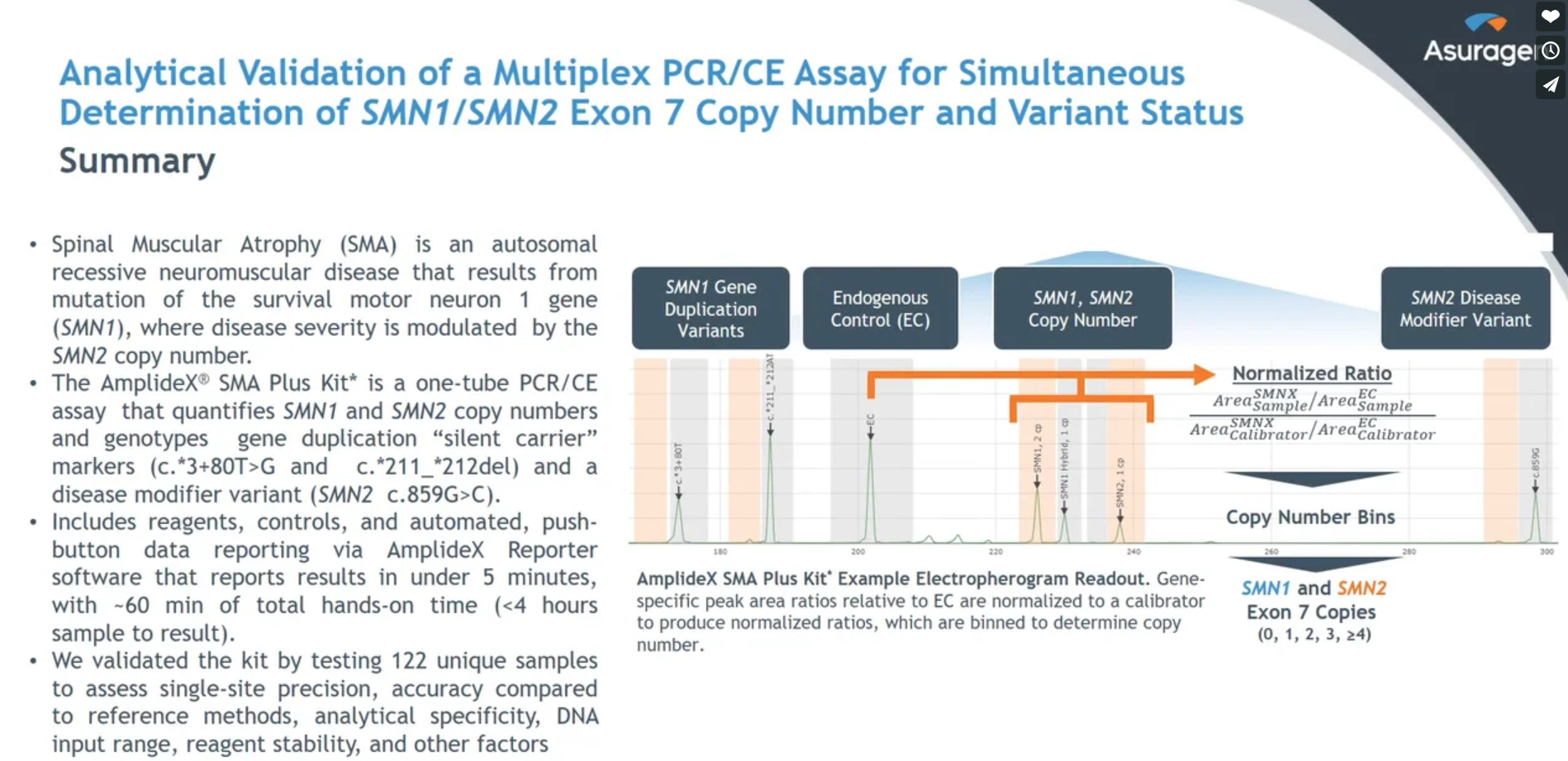
Analytical validation of a multiplex PCR/CE assay for simultaneous determination of SMN1/SMN2 exon 7 copy number and variant status
(Milligan J, et al).
(Click the image to access the video via Vimeo.)
Download a copy of the Milligan J, et al. poster slides.
Learn more:
Enquire about this article
"*" indicates required fields