From Antigens to Epitopes: Refining HLA-Antibody Risk Assessment
The wide adoption of virtual crossmatching to assess donor/recipient immunological risk has necessitated a focus on HLA-antibody definition beyond the traditional antigenic-level. Increased access to allelic-level donor and recipient HLA-typing, in combination with tools enabling greater definition of HLA-antibody profiles, can aid in more nuanced immunological risk assessment with the aim of optimising graft and patient outcomes [1]. Of note, this is of particular importance to highly sensitised patients where strategies such as ‘de-listing’ of HLA-antibody specificities may be required to gain access to transplantation.
It is well established that upon exposure to ‘non-self’ HLA, resultant HLA-antibodies are not directed against the entirety of the sensitising HLA-antigen but rather recognise polymorphic amino-acid residue clusters called functional epitopes or eplets [2]. These epitopes can be public (shared amongst multiple specificities) or private (unique to one specificity). Utilising epitope-analysis to ‘pick-apart’ HLA-antibody reactivity patterns can provide confidence in solid-phase assay results and guide antibody listing and compatibility decisions through greater understanding of cross-reactive potential. In recognition of this, the latest ‘British Society for Histocompatibility and Immunogenetics (BSHI) and British Transplantation Society (BTS) UK guideline on the detection of alloantibodies in solid organ (and islet) transplantation’, now suggests that the use of epitope analysis software may be of benefit in HLA-antibody profile interpretation [3].
The following short case studies provide examples of the utilisation of LABScreen™ Single Antigen ExPlex, One Lambda™ MagSort™ and additional online tools to help further define HLA-antibody profiles. LABScreen™ Single Antigen ExPlex provides increased breadth of allele coverage over the standard LABScreen™ Single Antigen panel which in turn broadens the number of patients who can benefit from allelic- and epitope-level HLA-antibody assessment. One Lambda™ MagSort™ is a panel of single HLA-antigen bound magnetic beads (59 antigen-specific bead sets across 6 locus-specific kits). They enable the isolation and elution of targeted HLA-antibodies from complex serum, thereby isolating antibodies of interest as well as supporting epitope-level HLA-antibody analysis.
Case 1 – Use of LABScreen™ Single Antigen ExPlex
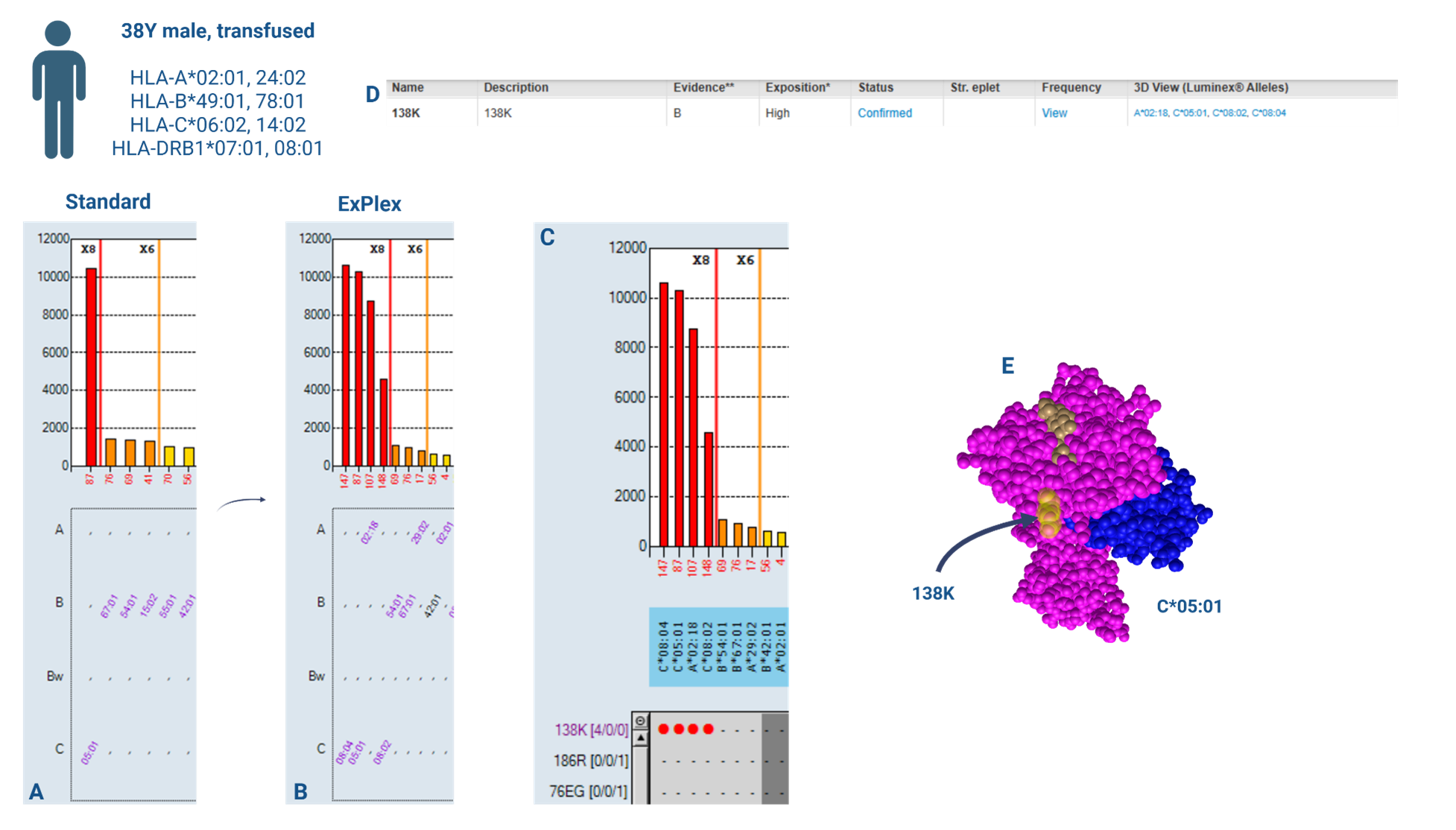
A previously transfused, 38-year-old male being worked-up for renal transplant was tested for HLA-antibodies using LABScreen™ Single Antigen Class I and II. No HLA-Class II antibodies were detected; however, a high-MFI HLA-C*05:01 antibody was present. No other antibodies were above local MFI thresholds for calling HLA antibody specificities (Figure 1A).
Due to the highly-specific nature of the profile, the result was queried as potentially non-specific serum interference and LABScreen™ Single Antigen Class I ExPlex reflex testing carried out. Allelic-level patient typing was available demonstrating HLA-C*05:01 was not ‘self’. In addition to HLA-C*05:01 reactivity, ExPlex testing showed strong reactivity for HLA-A*02:18, -C*08:02 and -C*08:04 beads (Figure 1B). With the ExPlex data and utilising the Epitope Panel available in HLA-Fusion™ 4.7.x (which draws from Duquesnoy’s HLA Matchmaker epitopes [4]), a clear epitope target of reactivity can be seen – 138K – which explains reactivity to these specificities in addition to the original HLA-C*05:01 (Figure 1C). For further confidence, the epitope was located on the HLA Eplet Registry [5] which serves as the main database of theoretical and confirmed HLA eplets recognisable by B-cell receptors (Figure 1D). The database lists 138K as an antibody-verified epitope. Utilising functionality within HLA Matchmaker, as part of HLA Fusion™ software, a 3D representation of the epitope was viewed providing visual confirmation of the epitope’s location on the surface of the molecule, again indicating antibody accessibility (Figure 1E).
In this case, the use of LABScreen™ Single Antigen ExPlex testing enabled confirmation of the presence of a high-level HLA*C*05:01 antibody, identified the responsible epitope and additional targets of reactivity to be considered for donor/recipient immunological risk assessment and listing as unacceptable antigens.
Figure 1: Utility of enhanced HLA-antigen coverage in identifying epitope targets of reactivity. A: Results of LABScreen™ Single Antigen Class I (standard panel) testing. B: Results of LABScreen™ Single Antigen Class I ExPlex testing. C: HLA Fusion™ 4.7.x Epitope Panel identification of epitope target of reactivity – 138K. D: HLA Eplet Registry record of 138K epitope. E: 3D visualisation of epitope location on HLA molecule.
Case 2: Use of One Lambda™ MagSort™
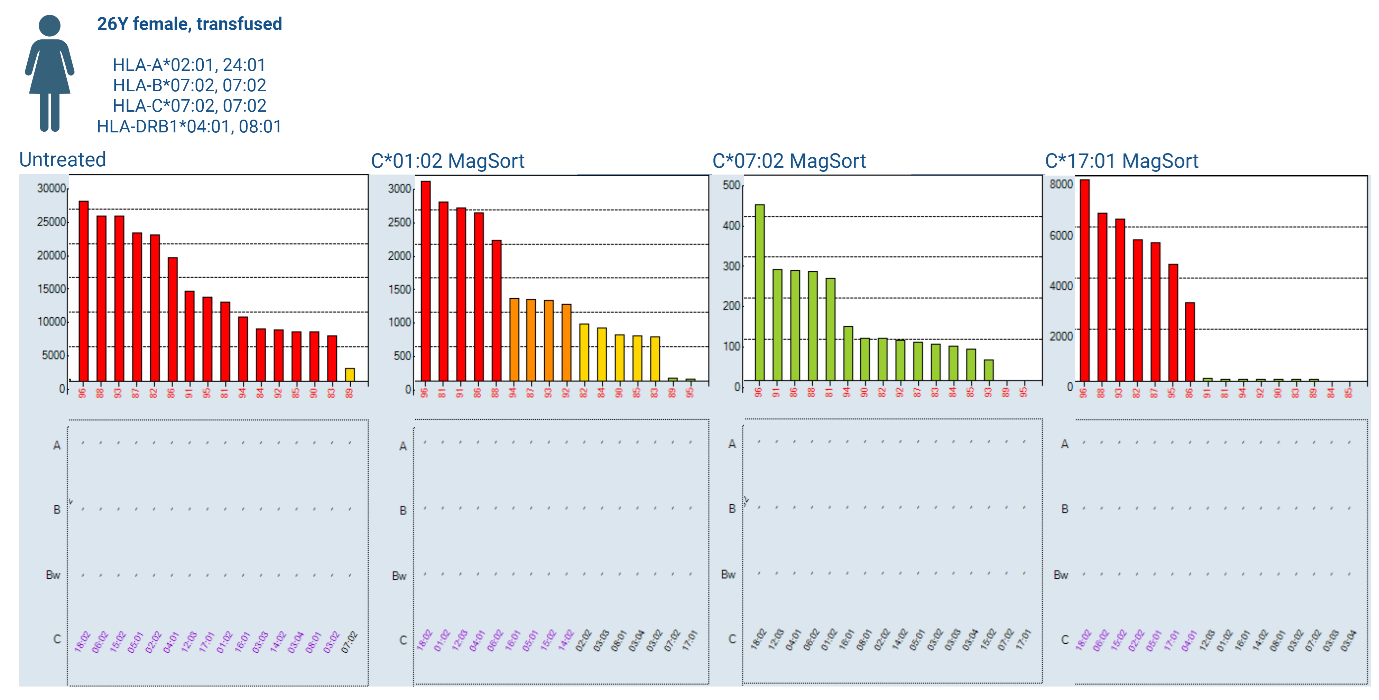
A previously transfused 26-year-old female, being worked up for transplant was tested for HLA-antibodies using LABScreen™ Single Antigen Class I and II. The HLA-Class I result was queried due to a largely pan-reactive HLA-C antibody profile (Figure 2; untreated). Further testing was felt necessary to confirm the profile due to low-level reactivity to ‘self’ as well as the impact to chance of transplant that listing the antibody profile as unacceptable would have for the patient.
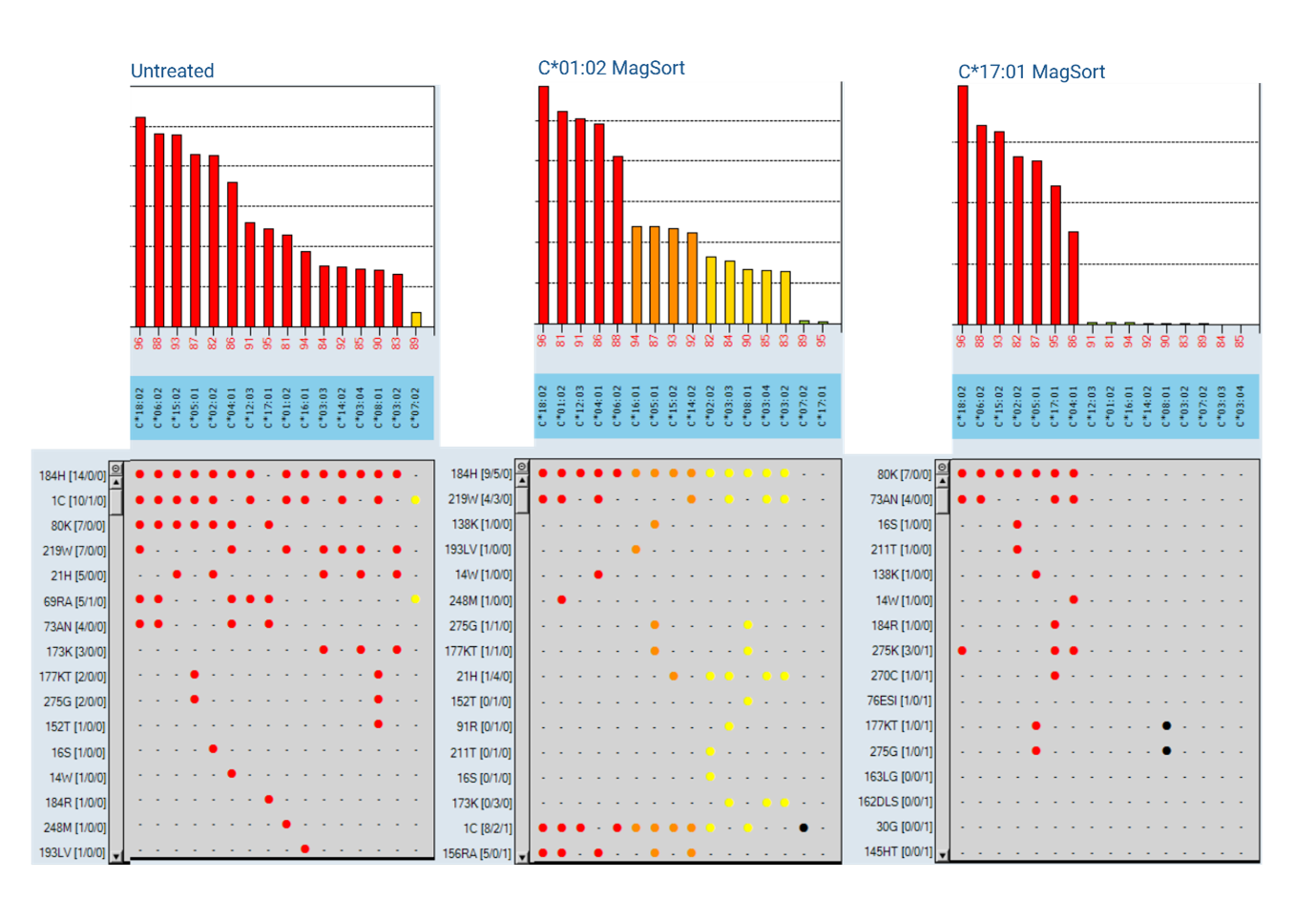
Three One Lambda™ MagSort™ beads (HLA-C*01:02, C*07:02 and C*17:01) were identified to isolate their respective antibodies from the primary sera. The HLA-C*07:02 MagSort™ bead (a ‘self’ antigen) did not isolate HLA-antibody from the sera (Figure 2; C*07:02 MagSort™). HLA-antibody was successfully isolated using the HLA-C*01:02 and -C*17:01 beads, each producing a distinct profile upon re-testing using LABScreen™ Single Antigen Class I (Figure 2; C*01:02 MagSort™ and C*17:01 MagSort™). Through isolating elements of the antibody profile and utilising the Epitope Panel functionality in HLA Fusion™ 4.7.x, again drawing from Duquesnoy’s HLA Matchmaker [4], confirmation of the antibody profile was obtained. It was possible to explain the full profile of reactivity (minus ‘self’) through reactivity to epitopes 184H and 80K (Figure 3), confirming an almost pan-HLA-C antibody profile. This testing approach provided the required reassurance to list the full profile as unacceptable antigens.
The use of One Lambda™ MagSort™ beads to isolate and elute antibodies removes the background properties associated with serum, enabling identification of non-specific serum interference in further solid-phase testing. In this case, providing confirmation that HLA-C*07:02 antibody was not present, as expected, due to the patient HLA-type. Pulling apart the reactivity profile through adsorption and elution was effective in identifying potential epitope targets of reactivity, confirming authenticity of the broad, clinically impactful antibody profile.
Figure 2: Pan HLA-C antibody profile detected by LABScreen™ Single Antigen Class I untreated and post-MagSort™ bead isolation (HLA-C*01:02, -C*07:02 and -C*17:01).
Figure 3: Epitope analysis of pan HLA-C antibody profile detected by LABScreen™ Single Antigen Class I untreated and post-MagSort™ bead isolation (HLA-C*01:02, -C*07:02 and C*17:01).
Webinar: Case Studies from the Epitope-Analysis Toolbox
These case studies demonstrate the utility of additional tools, both product and software features, in the HLA toolbox to facilitate epitope-based antibody analysis.
Watch our case presentation webinar to gain a better understanding of how One Lambda™ LABScreen Single Antigen ExPlex and One Lambda™ MagSort can be integrated into the analysis of complex (and surprising!) patient cases, and how epitope-based analysis tools in the HLA Fusion™ 4.7.x software can be used to facilitate antibody analysis.
Get in touch to request to view the webinar recording:
Contact the VH Bio team by filling in the form below for any questions around LABScreen™ Single Antigen ExPlex and One Lambda™ MagSort™ or for a demonstration of HLA-Fusion™ 4.7.x epitope-based analysis features (e.g. HLA Matchmaker and the Epitope Panel).
References
- Kardol-Hoefnagel T, Senejohnny DM, Kamburova EG, et al. Determination of the clinical relevance of donor epitope-specific HLA-antibodies in kidney transplantation. HLA. 2024;103(1):e15346. doi:10.1111/tan.15346
- Kramer CSM, Roelen DL, Heidt S, Claas FHJ. Defining the immunogenicity and antigenicity of HLA epitopes is crucial for optimal epitope matching in clinical renal transplantation. HLA. 2017; 90(1): 5-16. doi:10.1111/tan.13038
- BSHI & BTS UK guidelines on the detection of alloantibodies in solid organ (and islet) transplantation (2023). https://bts.org.uk/bshi-and-bts-uk-guideline-on-the-detection-of-alloantibodies-in-solid-organ-and-islet-transplantation/.
- Duquesnoy, Rene J. “A structurally based approach to determine HLA compatibility at the humoral immune level.” Human immunologyvol. 67,11 (2006): 847-62. doi:10.1016/j.humimm.2006.08.001
- Duquesnoy, R J et al. “16th IHIW: a website for antibody-defined HLA epitope Registry.” International journal of immunogeneticsvol. 40,1 (2013): 54-9. doi:10.1111/iji.12017
Enquire about this article
"*" indicates required fields