
Why Use Cell Lines to Study the Brain?
The brain is a highly complex organ and can develop equally complex diseases. A robust, cost-effective, and accessible way to investigate biological processes and disease mechanisms is through the use of cell lines, also referred to as in vitro models.
The brain controls essential functions such as movement, memory, learning, and decision-making. To do this, it relies on multiple specialised cell types, which broadly fall into two categories:
- Neurons, which transmit electrochemical signals throughout the brain and central nervous system (CNS)
- Glial cells, which provide structural, metabolic, and immunological support
Glial cells include astrocytes, oligodendrocytes, microglia, and ependymal cells, each with distinct roles. These cells are distributed across specialised brain regions such as the hypothalamus, cerebral cortex, brain stem, and the blood–brain barrier (BBB).
Cell line models allow researchers to study these cell types, their regional specialisation, and their role in disease using unlimited cell populations under controlled, reproducible conditions. These systems are also well suited to scalable applications such as drug screening.
Immortalised Neuronal Cell Lines from Cedarlane and VH Bio
Much of what is known about cell biology comes from cell culture models, including both primary cultures and immortalised cell lines. Each in vitro approach has advantages and limitations.
Immortalised cell lines are generally easier to culture and can be maintained for longer periods than primary cells. While they do not fully replicate the complexity of native brain tissue, they provide simplified, well-controlled models that allow researchers to focus on specific biological functions or disease mechanisms.
One common method of immortalisation involves retroviral transfer of the simian virus 40 T antigen (SV40 T-Ag) (Jung et al., 1994). SV40 T-Ag binds to the tumour suppressor proteins p53 and retinoblastoma (Rb), allowing cells to bypass normal cell-cycle checkpoints and continue proliferating. Unless stated otherwise, the neuronal cell lines described in this article have been immortalised using SV40 T-Ag.
VH Bio’s product partner, Cedarlane, offers a broad portfolio of neuronal and glial cell lines that express relevant neuropeptides, enzymatic markers, and biologically active receptors. These models support accurate in vitro assays for research into neurodegenerative disease, metabolic and developmental disorders, and many other areas of neurobiology.
Overview of Neuronal and Glial Cell Line Models
Hypothalamic Cell Lines
The hypothalamus is a small but vital brain region that regulates key physiological processes such as appetite, stress responses, circadian rhythms, and body temperature. Cedarlane provides a range of adult and embryonic hypothalamic cell lines derived from different hypothalamic regions, summarised in Table 1. These models include neuronal and supporting glial populations, such as astrocytes.
Table 1. Hypothalamic Cell Lines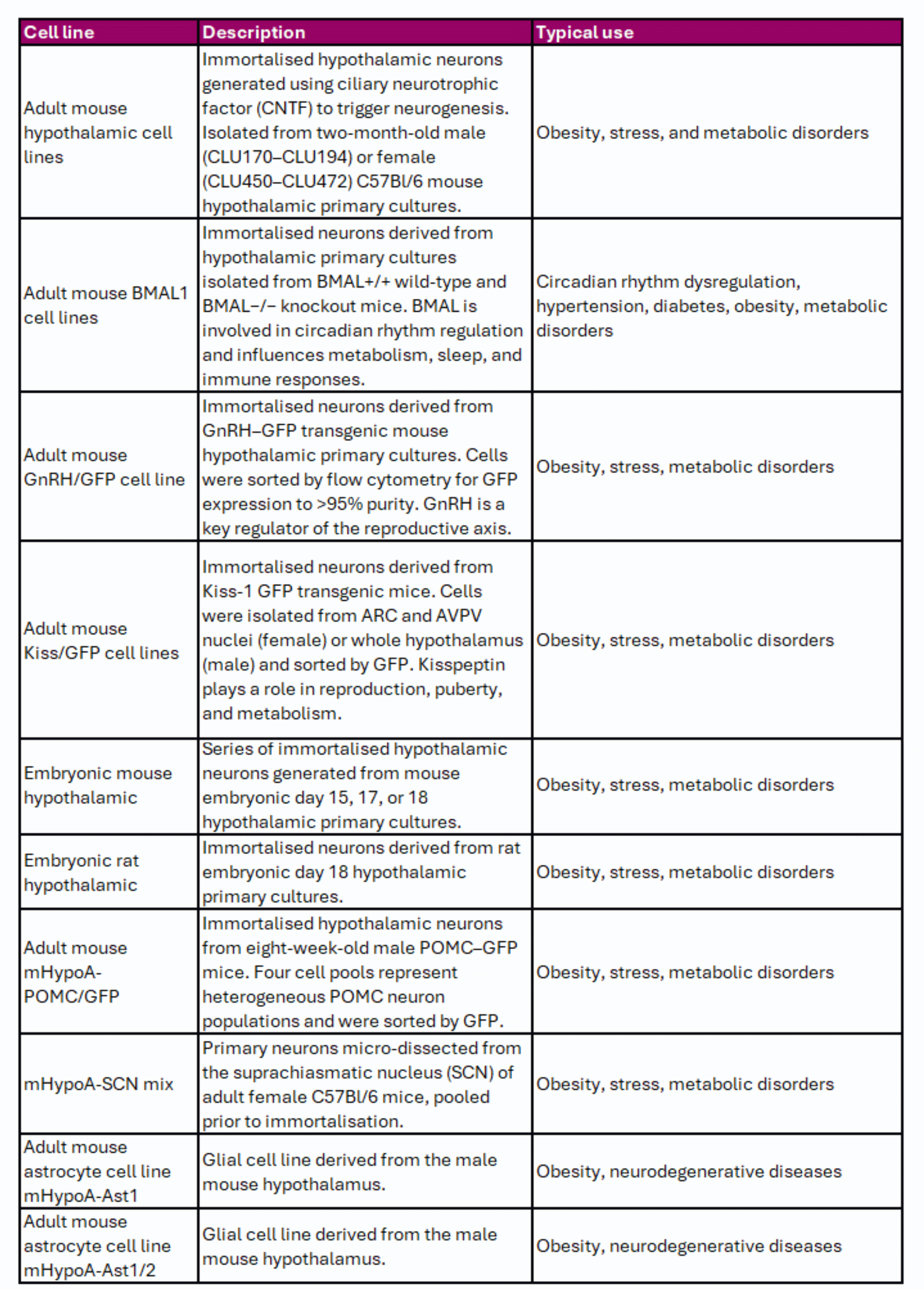
Oligodendrocytic Cell Lines
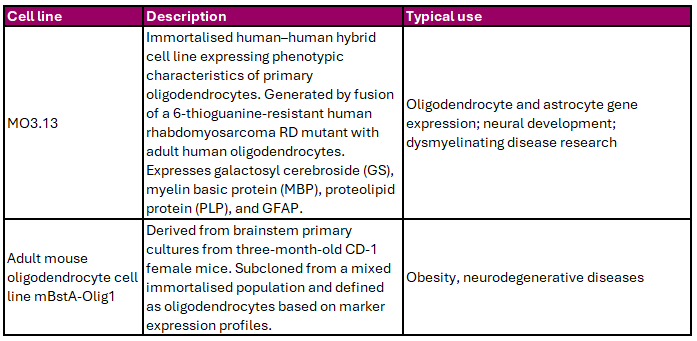
Oligodendrocytes produce the myelin sheath that insulates neuronal axons, enabling efficient signal transmission and providing metabolic support. Cedarlane offers oligodendrocyte-like cell lines that can be used to study myelination, glial biology, and neurodegenerative disease (Table 2).
Table 2. Oligodendrocytic Cell Lines
Hippocampal Cell Lines
The hippocampus plays a central role in learning, memory, and emotional regulation. Cedarlane supplies embryonic mouse hippocampal cell lines that support studies of neuronal development, maturation, and disease progression in a controlled in vitro environment (Table 3).
Table 3. Embryonic Hippocampal Cell Lines

Pituitary Gland Cell Lines
The pituitary gland is a key endocrine organ involved in hormone production and regulation of growth, metabolism, reproduction, blood pressure, and stress responses. Cedarlane offers adult-derived pituitary cell lines suitable for studies of hormone synthesis, secretion, and progenitor cell characteristics (Table 4).
Table 4. Pituitary Cell Lines

Endothelial Cell Line (Blood–Brain Barrier Model)
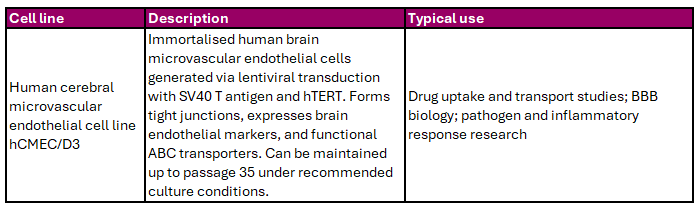
Endothelial cells form the structural basis of the blood–brain barrier (BBB), a tightly regulated interface that protects the CNS while allowing controlled molecular transport. The BBB model available from Cedarlane exhibits tight junction formation, endothelial marker expression, and functional ABC transporters (Table 5).
Table 5. Endothelial Blood–Brain Barrier Cell Line
Cerebral Cortex Glial Cell Line
The cerebral cortex is responsible for higher-order brain functions such as cognition, memory, and consciousness. Cedarlane provides an astrocyte cell line derived from the adult mouse cerebral cortex, suitable for studies of cortical glial biology (Table 6).
Table 6. Cerebral Cortex Glial Cell Line

Motor Neuron Cell Line
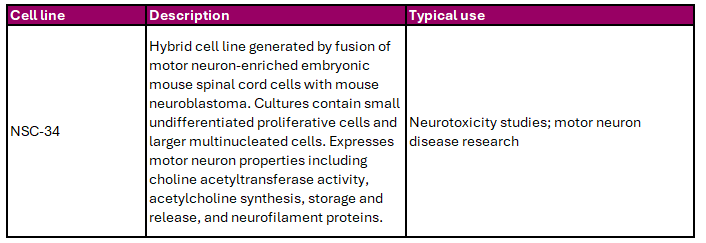
Motor neurons transmit signals from the brain to the peripheral nervous system to control muscle movement. Cedarlane offers the NSC-34 motor neuron-like cell line, summarised in Table 7, which is widely used in neurotoxicity and neurodegeneration research.
Table 7. Motor Neuron Cell Line
How to Choose, Maintain, and Use Neuronal Cell Lines
Choosing the Right Cell Line
Selecting an appropriate model begins with clearly defining your research question. Useful considerations include:
- Does the cell line represent the biology you want to study?
- Has it been used successfully in similar research?
- What are the known limitations of the model?
- Is the cell line readily accessible?
- Does it require specialised methods such as co-culture or 3D systems?
Maintaining Your Cell Line
Correct culture and maintenance are essential for preserving cell phenotype and experimental reproducibility. Key considerations include:
- Thawing procedures upon arrival
- Suitable culture medium and supplements
- Freezing early passage stocks
- Requirements for surface coatings or specialised cultureware
- Seeding density and passaging frequency
Detailed guidance is provided in individual cell line data sheets and by the supplier.
Validating and Using Your Cell Line
Before experimental use, cell lines should be characterised within the laboratory and compared with published literature. Questions to address include:
- What markers define the original phenotype?
- Does the phenotype change with passaging or confluence?
- Is differentiation required to induce relevant neuronal properties?
- Are published protocols available for differentiation or functional assays?
For technical questions or troubleshooting, users can contact VH Bio or the Cedarlane technical support team.
Why Use a Cell Line Model?
Cell line models provide a cost-effective, scalable, and reproducible alternative to in vivo approaches, particularly during early-stage research. They allow rapid hypothesis testing without the need for animal facilities or extensive training. Importantly, in vitro studies can generate mechanistic insights that guide downstream validation in animal models.
The following section highlights the NSC-34 cell line as an example of how neuronal cell line models can be applied in practice.
Spotlight: NSC-34 Motor Neuron Cell Line
The NSC-34 cell line is a widely used model for motor neuron biology and disease and is supplied by VH Bio on behalf of Cedarlane. It has been extensively characterised for morphology, differentiation, and functional behaviour.
Morphology, Characterisation, and Function
NSC-34 cultures contain two distinct cell populations: small, undifferentiated proliferative cells and larger, multinucleated cells. Upon differentiation, NSC-34 cells adopt neuron-like morphology, extend neurites, and form interconnected networks.
The cell line expresses key motor neuron markers, including choline acetyltransferase (ChAT), neurofilament triplet proteins, and components involved in acetylcholine synthesis, storage, and release. Electrophysiological studies show responses to neurotoxic agents affecting ion channels, cytoskeletal organisation, and axonal transport, supporting use in neurotoxicity and ALS research (Sabitha et al., 2016).
Origin and Limitations
NSC-34 is an immortalised hybrid cell line generated by fusion of motor neuron-enriched embryonic mouse spinal cord cells with mouse neuroblastoma cells. As a result, it cannot fully replicate the genetic and functional complexity of primary motor neurons. A documented limitation is reduced susceptibility to glutamate-mediated excitotoxicity compared with primary motor neuron cultures (Madji Hounoum et al., 2016).
Maintenance
NSC-34 cells are supplied at passage numbers typically ranging from 25 to 28. Thawing protocols recommend seeding directly into tissue culture plates without centrifugation, followed by a medium change after 4–6 hours to remove residual DMSO. Cells are maintained in DMEM supplemented with glucose, L-glutamine, sodium bicarbonate, and fetal bovine serum, with antibiotics added as required. NSC-34 cells adhere well to standard tissue culture plastic and do not require specialised surface coatings. While most studies use 2D cultures, 3D spheroid models have also been described (Arnaldi et al., 2024).
Differentiation
Differentiation protocols typically involve retinoic acid treatment on collagen-coated surfaces. Studies have shown increased neurite outgrowth, reduced proliferation markers, and enhanced expression of neuronal and synaptic proteins during differentiation (Maier et al., 2013). While differentiation enhances neuronal features, some functional limitations remain.
Disease Modelling Applications
NSC-34 cells are widely used to model amyotrophic lateral sclerosis (ALS) and other neurodegenerative conditions. Exposure to cerebrospinal fluid from ALS patients induces pathological changes such as neurofilament aggregation and reduced ChAT expression. The cell line has also been used to study neurotoxicity induced by metabolic stressors, including high fructose, and inflammatory cytokines such as TNF-α, supporting applications in drug screening and mechanistic research.
Summary
Neuronal and glial cell lines are powerful tools for advancing neurobiological research. Cedarlane, in partnership with VH Bio, offers a broad portfolio of well-characterised neuronal cell lines that support reproducible and scalable in vitro studies across a wide range of neurological research areas.
Contact us by filling in the form below to discuss your lab’s requirements or with any questions about our range of neuronal cell lines.
References
Arnaldi, P., Casarotto, E., Relucenti, M., Bellese, G., Cristina Gagliani, M., Crippa, V., Castagnola, P., Cortese, K. (2024) A NSC-34 cell line-derived spheroid model: Potential and challenges for in vitro evaluation of neurodegeneration. Microscopy Research and Technique. 87 (11) 2785-2800
Cashman, NR., Durham, HD., Blusztajn, JK., Oda, K., Tabira, T., Shaw, IT., Dahrouge, S., Antel, JP.(1992) Neuroblastoma x spinal cord (NSC) hybrid cell lines resemble developing motor neurons. Developmental Dynamics. 194 209-221
Jung, M., Crang, A., Blakemore, W., Hoppe, D., Kettenmann, H., Trotter, J. (1994) In vitro and in vivo characterisation of glial cells immortalised with a temperature-sensitive SV40 T antigen-containing retrovirus. Journal of Neuroscience Research. 37(2) 182-96
Kupershmidt, L., Weinreb, O., Amit, T., Mandel, S., Carri, M., Youdim, M. (2009) Neuroprotective and neuritogenic activities of novel multimodal iron-chelating drugs in motor-neuron-like NSC-34 cells and transgenic mouse model of amyotrophic lateral sclerosis. Federation of American Societies Experimental Biology Journal. 23(11) 3766-79
Lodha, D., Rajasekaren, S., Jayavelu, T., Subramaniam, J. (2022) Detrimental effects of fructose on mitochondria in mouse motor neurons and on C. elegans healthspan. Nutritional Neuroscience. 25(6) 1277-1286
Loganathan, N., Lieu, C., Belsham, D. (2024) Immortalization and Characterization of GFAP-expressing Glial Cells from the Adult Mouse Hypothalamus, Cortex, and Brain Stem. Neuroscience. 551 43-54 ASTROCYTE CELL LINE PAPER
Madji Hounoum, B., Vourc’h, P., Felix, R., Corcia, P., Patin, F., Guéguinou, M., Potier-Cartereau, M., Vandier, C., Raoul, C., Andres, C., Mavel, S., Blasco, H. (2016) NSC-34 Motor Neuron-Like Cells Are Unsuitable as Experimental Model for Glutamate-Mediated Excitotoxicity. Frontiers in Cellular Neuroscience. 10 (118) doi: 10.3389/fncel.2016.00118
Maier, O., Böhm, J., Dahm, M., Brück, S., Beyer, C., Johann, S. (2013) Differentiated NSC-34 motorneuron-like cells as experimental model for cholinergic neurodegeneration. Neurochemistry International. 62 1029-1038
Sabitha, K., Sanjay, D., Raju, T., Laxmi, R. (2016) Electrophysiological characterisation of NSC-34 cell line using microelectrode array. Journal of the Neurological Sciences. 370 134-139
Enquire about this article
"*" indicates required fields
