
Real-World Performance Backed by Peer-Reviewed Data
Clinical laboratories and research institutions are under increasing pressure to deliver molecular diagnostic results quickly and accurately. The geneLEAD VIII automated sample-to-result RT-PCR platform addresses this need, combining automation with robust analytical performance across multiple applications.
In this blog, we’ll highlight key findings from recent peer-reviewed studies demonstrating how the geneLEAD VIII performs in practice and demonstrate why the system is an impactful solution for molecular diagnostics:
The geneLEAD VIII: Key Findings from Independent Studies
The geneLEAD VIII platform from Precision System Science (PSS) is a system that uses magnetic bead-based nucleic acid extraction. It is unique in its ability to integrate nucleic acid extraction and real-time PCR in a single automated workflow. It does this directly from clinical samples, all within a single instrument.
The studies found that the geneLEAD VIII demonstrate both analytical performance and workflow efficiencies:
- Detection limits comparable to manual RT-PCR methods
- Approximately 3 hours 40 minutes reduction in turnaround time
- 46 minutes less hands-on time per four samples
Learn about the studies and the results achieved with the GeneLead VIII:
Multiplex Respiratory Virus Detection: High Sensitivity and Strong Concordance
A 2025 study evaluated the performance of geneLEAD VIII coupled with the VIASURE SARS-CoV-2, Flu & RSV Real-Time PCR Detection Kit to detect four major respiratory pathogens: SARS-CoV-2, influenza A (IAV), influenza B (IBV), and respiratory syncytial virus (RSV).
Key takeaways from this clinical performance evaluation include:
Sensitive viral detection limits: Using virus-spiked clinical matrices (saliva and nasal swabs), the platform detected SARS-CoV-2 down to 10⁰ TCID₅₀/mL, IAV to ≤10⁻² TCID₅₀/mL, IBV to 10⁰ TCID₅₀/mL, and RSV to 10² TCID₅₀/mL. These limits are comparable with — and in some cases more sensitive than — traditional manual RT-qPCR assays.
High analytical concordance: When comparing Ct values between geneLEAD VIII and manual assays, Pearson correlation coefficients were robust across targets: 0.90 for SARS-CoV-2, 0.85 for IAV, 0.92 for IBV, and 0.95 for RSV — indicating strong agreement in quantitative performance.
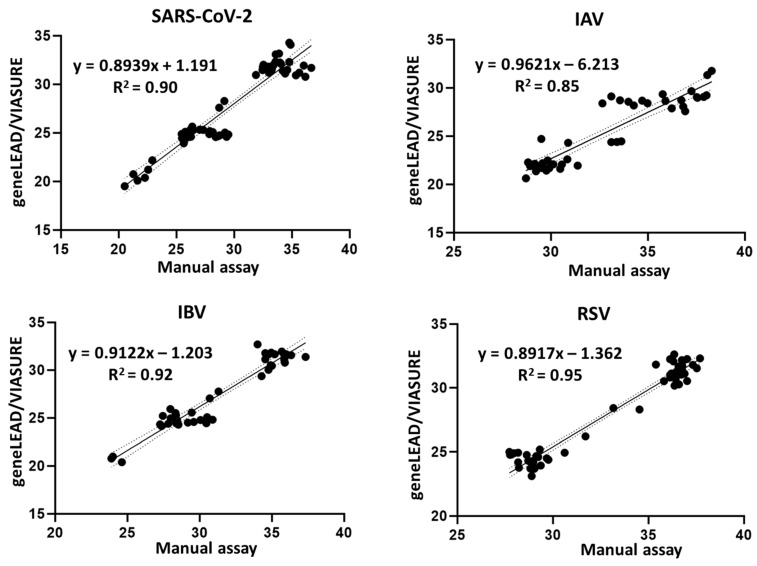
Figure 1. Simple linear regression plots of Ct values for the detection of SARS-CoV-2 (n = 62), IAV (n = 44), IBV (n = 41), and RSV (n = 56), obtained using geneLEAD/VIASURE versus the manual assays. Each plot displays the equation for the best-fit line and Pearson’s coefficient of determination (R2). The dotted lines indicate the 95% confidence bands of the best-fit line.
Effective in co-infection models: geneLEAD VIII maintained stable Ct performance even in multiple-infection simulations, underscoring its utility in complex clinical scenarios.
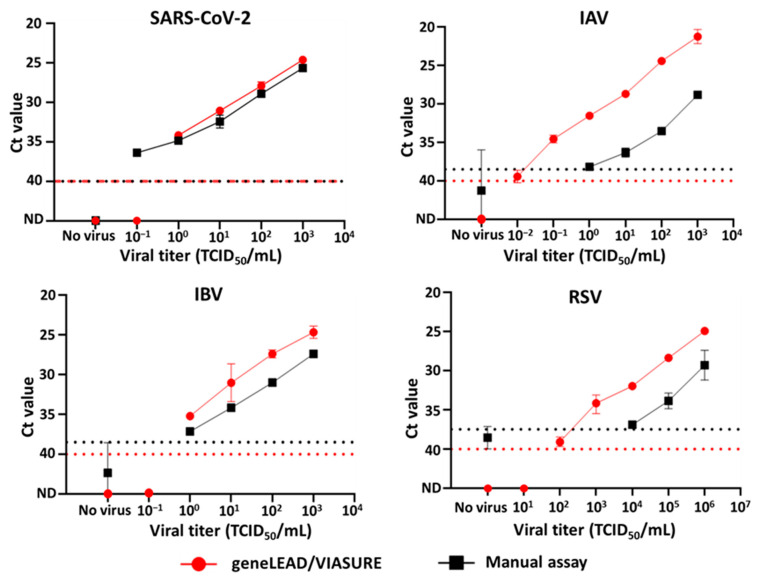
Figure 2. Comparison of the viral titer detection limits of SARS-CoV-2 (n = 2), IAV (n = 2), IBV (n = 2), and RSV (n = 2–4) between analyses conducted using geneLEAD/VIASURE (●) and the manual assays (■). The red and black dotted lines denote the cutoff Ct values for the automated system and the manual assays, respectively.
These results support geneLEAD VIII’s analytical accuracy and operational compatibility with standard RT-PCR benchmarks.
100% Sensitivity and Specificity for Rapid Detection of Antimicrobial Resistance Genes
Beyond virus detection, geneLEAD VIII also delivers strong performance for antibacterial resistance diagnostics. In a separate 2025 study, researchers evaluated its use for detecting vancomycin resistance genes (vanA and vanB) in Enterococcus isolates – genes responsible for vancomycin resistance in Enterococcus (VRE) – which is clinically important in hospital infections. The research directly compared the platform to a validated in-house multiplex PCR.
Study highlights include:
Sensitivity and specificity: geneLEAD VIII achieved 100% sensitivity and 100% specificity for both vanA and vanB targets when tested across 200 clinical isolates.
Efficient workflow: Compared to the reference PCR workflow, the system reduced turnaround time by approximately 3 hours and 40 minutes and decreased hands-on preparation time by nearly 46 minutes per four samples.
Conclusion: Streamlined Operations and Laboratory Impact
The studies demonstrate that the key benefits of geneLEAD VIII include:
- Reduced manual labour and error risk through hands-free automation
- Parallel sample processing: multiple samples can be run in a single workflow with consistent quality
- Scalability for diverse testing needs, from respiratory panel diagnostics to targeted resistance gene assays
What This Means for Your Lab
The geneLEAD VIII system offers robust, reliable molecular diagnostics backed by independent performance studies. Its high sensitivity, broad pathogen detection, rapid turnaround, and fully automated workflow help clinical laboratories and research institutions to optimise RT-PCR testing while saving time.
For labs looking to streamline diagnostics and improve patient care, the platform represents a technically proven and versatile solution.
Get in touch by filling in the form below to discuss how the geneLEAD VIII can integrate into your workflow.
Learn more:
References
Komu, J. G., Jamsransuren, D., Matsuda, S., Ogawa, H., & Takeda, Y. (2025). Performance Evaluation of a Fully Automated Molecular Diagnostic System for Multiplex Detection of SARS-CoV-2, Influenza A/B Viruses, and Respiratory Syncytial Virus. Diagnostics (Basel, Switzerland), 15(14), 1791. https://doi.org/10.3390/diagnostics15141791
Mori, K., Matsumura, Y., Tsuda, Y., Shinohara, K., Tsuchido, Y., Yamamoto, M., & Nagao, M. (2025). Automated molecular detection of the vancomycin resistance genes vanA and vanB using the geneLEAD VIII platform. Access microbiology, 7(8), 001044.v4. https://doi.org/10.1099/acmi.0.001044.v4
Enquire about this article
"*" indicates required fields
