
Intro
In the field of transplantation, antibody detection strategies have evolved significantly over the last 20 years. Some of us remember using a plethora of methods to detect and define alloreactive antibodies: flow cytometry of patient sera against pooled cell lines, HLA ELISA such as the Lambda Antigen Tray™, and reading many, many complement-dependent cytotoxicity (CDC) trays (in-house and commercial, fresh cells and frozen, and random versus selected donors). The process was laborious and often limited in the ability to define antibody specificities in highly-sensitised patients.
One Lambda™ revolutionised alloantibody detection with the introduction of bead-based solid phase assays: initially flow cytometry-based (FlowPRA™) and latterly using Luminex® xMAP® technology which allowed the multiplexing of up to 100 analytes in a single assay. The LABScreen™ product range was introduced over 25 years ago: a suite of products allowing for the detection of HLA and MICA-specific antibodies (LABScreen™ Mixed) and the definition of antibodies to individual HLA specificities (LABScreen™ Single Antigen). LABScreen™ PRA holds a unique position: sometimes used instead of LABScreen™ Mixed for HLA antibody detection, a standardised assay for defining percentage panel-reactive antibody (%PRA), and able to define HLA specificities in moderately-sensitised individuals. LABScreen™ PRA is also a valuable reflex test: ‘reflexing up’ to confirm Undetermined LABScreen™ Mixed results and ‘reflexing down’ to substantiate antibody specificities determined using LABScreen™ Single Antigen.
LABScreen™ products have become the gold standard for alloantibody detection, however a suite of additional tools have been developed in recent years to complement and refine alloantibody detection and definition.
LABScreen™ Single Antigen Supplement and ExPlex: the ‘Extended Panels’.
LABScreen™ Single Antigen was developed within the confines of ‘real estate’, with xMAP® Technology limited to 100 analytes when using the CE-marked Luminex® 100™/200™ (LABScan™ 100) instruments. For LABScreen™ Single Antigen this was sufficient coverage to define antibodies to all classical HLA phenotypes (‘serological specificities’). LABScreen™ Single Antigen Supplement was released thereafter as an ‘add-on’ panel (to be run as a separate assay) in response to requests for additional antigen targets.
Launch of the FLEXMAP 3D® (LABScan3D™, an advanced multiplex analyser) now allows for the multiplexing of up to 500 analytes in a single assay. LABScreen™ Single Antigen ExPlex in many ways mirrors Supplement, however an important difference is the ability to combine the HLA Class I ExPlex assay in the same well as the classic Single Antigen HLA Class I assay; similarly for HLA Class II. This allows laboratories to maintain the same workflow and throughput whilst obtaining enhanced HLA antibody data and the resulting benefits. Conversely Single Antigen Supplement and ExPlex can be run separately and data combined with classic Single Antigen subsequently, perhaps allowing for a more ‘targeted’ approach to use.
The ’Extended Panels’ increase HLA Class I coverage from 97 to 151 antigens, and HLA Class II coverage from 95 to 119 antigens.
- Additional common HLA alleles are covered, e.g. HLA-A*02:05, HLA-C*07:01, HLA-DRB1*13:02, HLA-DQB1*05:03.
- The diversity of HLA alleles is greater, contributing to health equity. For example, the inclusion of HLA-A*02:07, HLA-DRB1*04:06 and HLA-DQB1*03:19 means the Extended Panels are more representative of the global population.
- Together the Extended Panels contribute an additional 60 Common and 18 Well-Documented alleles (Mack et al., 2013).
Given the classic Single Antigen panels provide coverage for all serological specificities, why do we need more antigens? Antibodies are not raised to individual HLA specificities (e.g. HLA-A2) but rather to epitopes shared between HLA specificities. This knowledge is not new (e.g. the ‘public’ epitope on HLA-A28 versus the ‘private’ epitopes on HLA-A68 and HLA-A69), however our understanding of epitopes has evolved over time: beyond Rodey’s cross-reactive group (CREG) classification (Rodey et al., 1994) to more refined structurally-based algorithms that help define epitopes at the molecular level (e.g. Duquesnoy’s eplets and HLA Matchmaker algorithm, 2006). The benefits of including more antigens are many:
- The Extended Panels provide a greater depth and breadth of antigen coverage, providing more datapoints and thus more confidence when analysing complex antibody profiles (Figure 1).
- The Extended Panels provide a greater depth and breadth of epitope coverage, providing more datapoints to facilitate epitope-based analysis (e.g. using HLA Matchmaker) (Figure 2).
- Epitope-based algorithms are incomplete; novel epitopes continue to be defined. More datapoints facilitate epitope-based analysis at the molecular level.
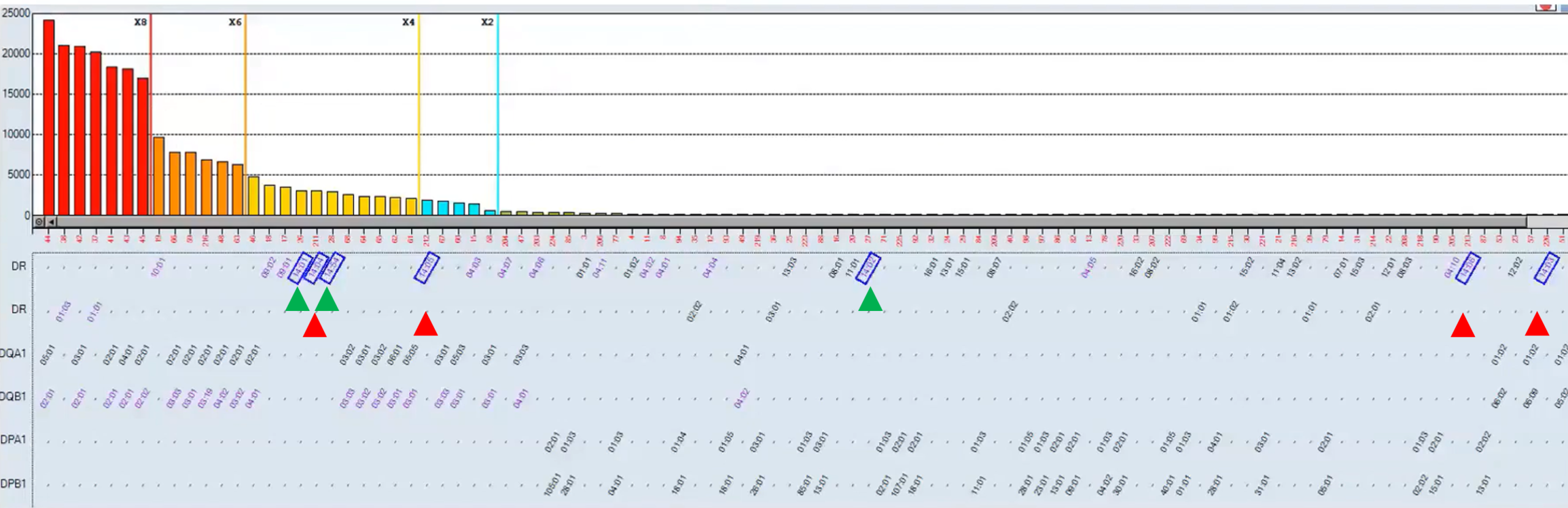
Figure 1. HLA-DR14 reactivity is seen using the classic Single Antigen panel (green) and the Single Antigen ExPlex panel (red); HLA Matchmaker analysis demonstrates a probable antibody to the 70R eplet.
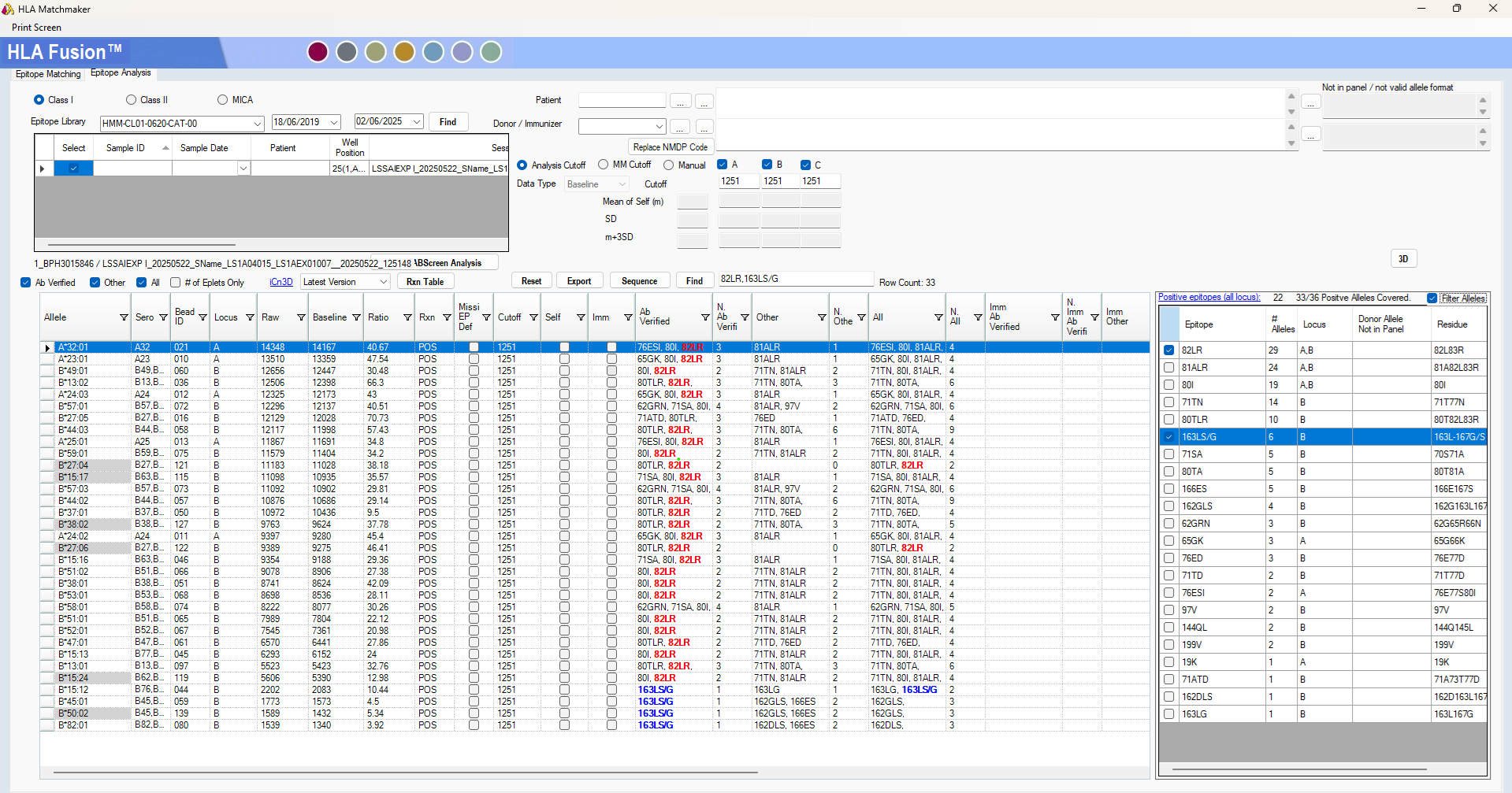
Figure 2. HLA Matchmaker analysis supports a HLA-Bw4-specific antibody (82LR eplet) amongst other eplet specificities. Single Antigen ExPlex allelic specificities are shaded grey.
The inclusion of additional antigens should not disadvantage patients (i.e. by defining more antibodies) but instead help refine antibody analyses. Consider epitope-based analysis to help refine cutoffs in Single Antigen-based assays, and additional HLA Class II antigens facilitating the definition of alpha-chain versus beta-chain antibodies. Similarly, comprehensive antibody definition is essential in the era of virtual crossmatching. A study by Jani et al. (2017) demonstrated discrepancies between flow cytometry and virtual crossmatching were due to ‘allelic HLA antibodies’ in 4.5% of cases, whilst Zavyalova et al. (2021) reported 15% patients awaiting transplantation have allele-specific anti-HLA antibodies. Examples of allele-specific antibodies have been seen in external quality assurance (EQA) sera (Figure 3) and are supported by real-life examples (Figure 4) and case studies. Hence, the value of extended Single Antigen panels in HLA-specific antibody definition are becoming clear with continuing reports and ongoing studies providing a firm evidence base to support their utility.
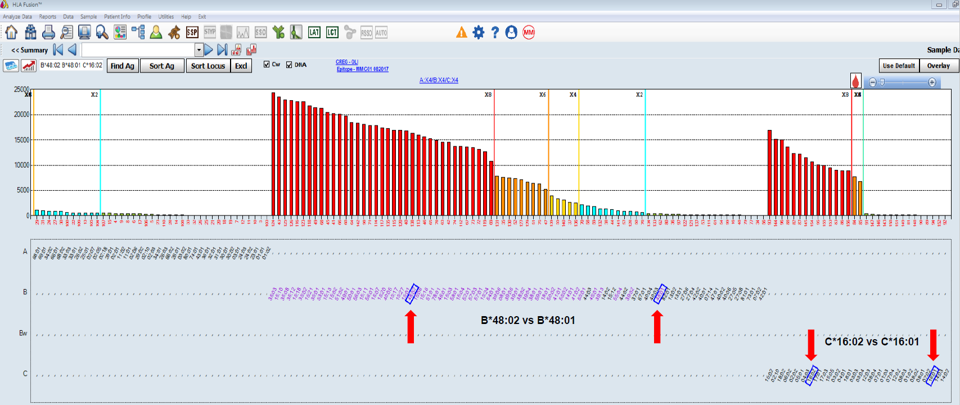
Figure 3. HLA antibodies are evident to HLA-B48 (HLA-B*48:02) and HLA-Cw16 (HLA-C*16:02) only by including the Single Antigen ExPlex panel.

Figure 4. Inclusion of the Single Antigen ExPlex panel identifies a DSA to HLA-DRB1*13:02; explained by an antibody to the 37N eplet.
One Lambda™ MagSort™
One Lambda™ MagSort™ is effectively a panel of HLA antigens conjugated to magnetic beads, akin to LABScreen Single Antigen™. MagSort™ allows antibody(ies) to a specific HLA antigen to be isolated from polyclonal serum in a targeted manner, allowing for a more focused dataset. The assay results in two endpoints: adsorbed serum (with the specific antibody(ies) depleted), and antigen-specific antibody(ies) that are eluted from the MagSort™ bead. MagSort™ is a flexible research tool with many applications still to be defined, and provides standardisation relative to alternative cell-based approaches to adsorption/elution (Liwski et al., 2022).
Figure 5 demonstrates MagSort™ in action. A complex antibody profile from a highly-sensitised patient is treated using a HLA-A*02:01 MagSort™ bead. The adsorbed serum and eluate is then further tested using LABScreen™ Single Antigen and ExPlex: selected specificities are reduced (adsorbed serum) with the eluate showing reactivity to HLA-A2 antigens, HLA-A28 (HLA-A68 and HLA-A69), and additional reactivity to e.g. HLA-B17 (HLA-B57 and HLA-B58). The pattern of reactivity can be analysed at the epitope-level to define a probable epitope specificity(ies).
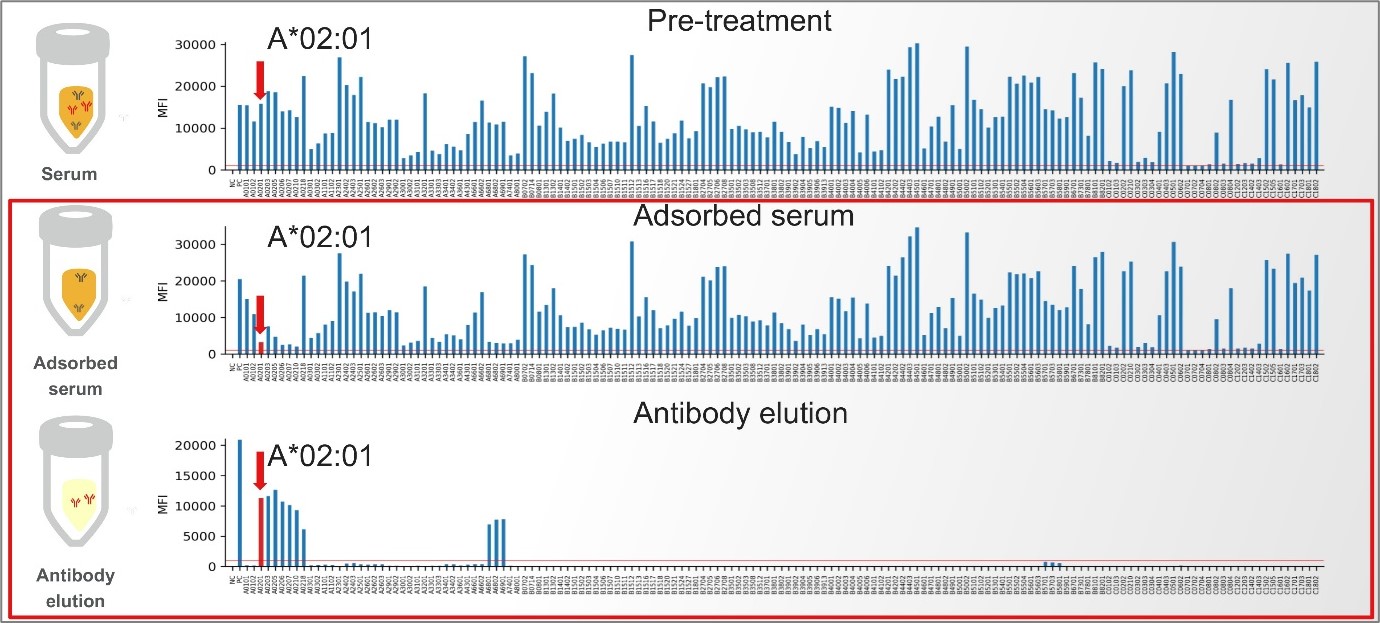
Figure 5. One Lambda™ MagSort™ helps define a cross-locus antibody to HLA-A2, HLA-A28 and HLA-B17.
At the practical level a clear application of MagSort™ is to facilitate epitope-specific antibody definition. Data can be used to explain patterns of HLA-specific reactivity; the output of this could be e.g. more comprehensively defined serological specificities (and Unacceptable [mismatched] Antigens in a potential donor). Conversely, ‘groups’ of serological specificities (Unacceptable Antigens) can be defined to aid delisting strategies and facilitate transplantation in highly-sensitised patients. An alternative application could be the use of defined-MFI, mono/minimally-specific sera to inform flow cytometry crossmatch cutoffs. The opportunities are seemingly endless.
HLA Fusion™
Innovation doesn’t always occur at the product-level, but rather within software-driven analysis. HLA Fusion™ is used for the analysis of One Lambda’s IVD-certified portfolio, whilst HLA Fusion™ Research software analyses RUO products such as LABScreen™ Autoantibody.
The LABScreen™ module within HLA Fusion™ has undergone significant enhancements over time: features such as patient (self) antigen and donor antigen flagging facilitate cut-off definition and the assignment of donor-specific antibodies (DSA), the CREG table and CREG map aid antibody analysis, and specific antibodies can be tracked longitudinally using the Antibody Tracking function. HLA Matchmaker has been integrated into HLA Fusion™ since 2018 (v4.2), circumventing epitope-based analysis in Excel and with continued improvements, e.g. the colour-coding of eplet specificities.
HLA Fusion™ v4.7.x brought significant enhancements to epitope-based analysis. The Eptiope Panel integrates epitope analysis with the LABScreen histogram, providing a highly visual aid to epitope-based analysis and epitope-specific antibody definition (in contrast to the table-based HLA Matchmaker module) (Figure 6). Further, the acknowledgement that not all HLA epitopes are defined has led to inclusion of the Amino Acid module where HLA antibody data are analysed against linear amino acid sequences to better explain patterns of antibody reactivity and facilitate novel epitope characterisation.

Figure 6. Splitting epitopes present on HLA Class II alpha and beta chains, the Epitope Panel helps define antibodies with accuracy.
Innovation in HLA-specific antibody definition, facilitated by advanced multiplexing (FLEXMAP® 3D / LABScan3D™), extended LABScreen™ Single Antigen panels (Supplement and ExPlex), One Lambda™ MagSort™ and HLA Fusion™-driven analysis is supporting a new era in transplant diagnostics where epitope-based antibody analysis can be implemented with ease and with increased confidence. The Team at V.H. Bio Limited are available to support you through product and software demonstrations, and by providing and signposting valuable learning resources.
Non-HLA: LABScreen™ Multi and Autoantibody
HLA is not the only alloantigen thought to be important in transplantation. In the mid-2000s antibodies to the polymorphic Major Histocompatibility Complex class I chain-related A (MICA) antigen were studied in the context of solid organ transplantation (hence inclusion in the LABScreen™ Mixed and LABScreen™ MICA Single Antigen panels), however opinion remains divided as to the clinical significance of this alloantigen.
LABScreen™ Multi
The importance of antibodies to Human Neutophil Antigens (HNA) is well established in granulocyte immunology and transfusion, particularly as a cause of Transfusion-Related Acute Lung Injury (TRALI). As a result One Lambda™ released LABScreen™ Multi for the simultaneous detection of HLA antibodies and the identification of HNA antibodies, in a single assay. LABScreen™ Multi is analogous to LABScreen™ Mixed, with the MICA panel swopped for variants of HNA-1, HNA-2, HNA-3, HNA-4 and HNA-5. LABScreen™ Multi follows the standard LABScreen™ workflow (with centrifugation-based wash steps) and is similarly IVD-certified.
More recently the importance of anti-HNA antibodies in solid organ transplantation has been recognised (Key et al., 2019). Specifically, anti-HNA-3A antibodies can result in positive flow cytometry crossmatch and are associated with graft pathology. ca. 5% of the (Caucasian) population are HNA-3B,3B and so at risk of developing antibodies to HNA-3A: often parous females, although anti-HNA-3A antibodies have also been reported in males (Docker et al., 2024). LABScreen™ Multi is an effective tool to screen transplant recipients for both HLA and HNA antibodies simultaneously.
LABScreen™ Autoantibody
Like MICA and HNA, examples of antibodies to non-HLA targets exist in the literature, often with associated pathology. Examples include anti-vimentin antibodies in cardiac and renal transplantation (Zhang et al., 2017), and the association of anti-PLA2R antibodies with renal transplant outcomes (Kanigicherla et al., 2013). The Banff classification system provides a standardised nomenclature for classifying kidney transplant pathology; the importance of non-HLA antibodies is such that their occurrence should be considered (Loupy et al., 2017).
One Lambda™ developed LABScreen™ Autoantibody with consideration of the literature: of the 39 non-HLA antigens, 25 were included due to citations in the transplant literature. These include Angiotensinogen (kidney), Regenerating islet-derived protein 3-alpha (pancreas), Myosin (heart), Tubulin alpha-1B chain (lung) and Glutathione S-transferase theta-1 (haematopoietic stem cell). The LABScreen™ Autoantibody panel is split across three panels and largely follows the standard LABScreen™ protocol, with Group 3 (Collagen and Fibronectin proteins) requiring a minor modification (due to the ‘sticky’ nature of these matrix proteins).
LABScreen™ Autoantibody is a RUO assay designed to facilitate research in the field of non-HLA immunity and is analysed using the HLA Fusion™ Research software.
Enhanced Crossmatching: FlowDSA-XM™
Historically, clinical transplantation was built upon three diagnostic pillars:
- Antibody detection and definition;
- Genotyping; and
- Recipient-donor crossmatching.
The physical recipient-donor crossmatch has somewhat diminished with the introduction of the virtual crossmatch to assess patient-donor compatibility from robust recipient antibody definition and donor genotyping. In many laboratories the CDC crossmatch (CDCXM) has been replaced entirely by flow cytometry crossmatching (FCXM), and crossmatching may be performed pre-transplant (e.g. for living donor transplants), peri-transplant (e.g. for highly-sensitised patients allocated a deceased donor organ) or post-transplant (e.g. to risk-assess the impact of DSA).
FlowDSA-XM™ assay occupies a unique place in the One Lambda portfolio, contributing to the third pillar of recipient-donor crossmatching. The FCXM typically combines recipient sera with donor lymphocytes, with T- and B-cell differentiation indicating whether antibodies are directed towards HLA Class I (T- and B-cells) or HLA Class II (B-cells). Interpretation of the FCXM can be confounded by reactivity to non-HLA targets (which may or may not be clinically significant), high-background or non-specific reactivity, immunosuppression (e.g. Rituximab) etc.
The FlowDSA-XM™ parallels the FCXM: recipient serum is incubated with donor lymphocytes, which are subsequently lysed and HLA-antibody complexes are ‘captured’ and labelled before reading on a flow cytometer (Figure 7). The Capture Beads split into three populations representing HLA Class I, HLA Class IIa and HLA Class IIb, with Median Channel Shift (MCS) against each gated population indicating binding of HLA-specific antibodies.
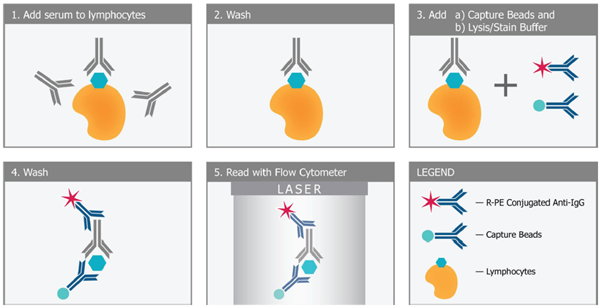
Figure 7. Principle of the FlowDSA-XM™ assay.
FlowDSA-XM™ builds on the sensitivity of the FCXM and ability to detect non-complement fixing antibodies (cf. the CDCXM) through the selective detection of HLA-specific antibodies. The workflow is familiar, fresh or frozen lymphocytes from various sources can be used, and the length of testing is similar to the standard FCXM. FlowDSA-XM™ is yet another tool offered by One Lambda to facilitate transplantation, and sits alongside the LABScreen™ assays (including those identifying non-HLA targets) and One Lambda™ MagSort™ to better predict and interpret recipient-donor compatibility, facilitating transplantation and supporting optimal outcomes.
Get in touch
V.H. Bio Limited have an excellent team to support our customers in the UK, Ireland and Barbados; get in touch if you want to know more about any of the ‘new’ One Lambda™ products available to better define alloantibodies. If you didn’t realise these excellent products were available, make sure you’re on our mailing list and follow us on LinkedIn and X.
References.
Docker et al. (2004) Int J Immunogenetics. 51(S2):55.
Duquesnoy et al. (2006) Front Immunol. 13(8):434.
Jani et al. (2017) Hum Immunol. 78(2): 72.
Liwski et al. (2022) Front Genet. 30(13): 1059650.
Loupy et al. (2017) Am J Transplant. 17(1): 28.
Kanigicherla et al. (2013) Kidney International. 83(5): 940.
Key et al. (2019) JRTS. 2(2): 81.
Mack et al. (2013) Tissue Antigens. 81(4): 194.
Rodey et al. (1994) Hum Immunol. 39(4): 272.
Enquire about this article
"*" indicates required fields