
Watch: Rapid, Comprehensive, and Complete SMN1/2 Profiling with the AmplideX® SMA Plus Kit (CE-IVD)
Asuragen recently showcased their latest product developments via a live corporate satellite and scientific poster presentation at the European Human Genetics Virtual Conference ESHG 2020.2, which ran from June 6-9. The videos [...]

Webinar – Broad SMN1 and SMN2 profiling with the AmplideX® PCR/CE SMN1/2 Plus Kit
Looking Beyond Copy Number: Broad SMN1 and SMN2 profiling in under four hours with the AmplideX® PCR/CE SMN1/2 Plus Kit (RUO) Spinal muscular atrophy (SMA) is a devastating neurodevelopmental illness caused by a loss of SMN1 copies with disease severity [...]

Webinar – The QuantideX® qPCR BCR-ABL IS Kit: A BCR-ABL Monitoring System that Meets and Exceeds Clinical Expectations for Testing
Clinical expectations for BCR-ABL monitoring have evolved over time both in response to more sensitive tests being developed and new treatments and management approaches emerging. Largely in response to these medical advances, many [...]

How the QuantideX® qPCR BCR-ABL IS Kit Provides a Simple, Sensitive, and Scalable BCR-ABL Monitoring Solution for a Laboratory
To mark the launch of the ground-breaking Quantidex® QPCR BCR-ABL IS Kit, manufacturer Asuragen held a series of three webinars. We covered the first two webinars here and here, so head there [...]

AllType™ FASTplex™ – Combining Multiplex PCR with Workflow Simplicity
Whilst most molecular HLA typing methodologies remain relatively static, next-generation sequencing (NGS) has tended to evolve quickly as product manufacturers respond to user suggestions, reagent development and changes in sequencing technology. One [...]

Analytical Validation of a BCR-ABL1 Monitoring System that Surpasses Current Testing Requirements
To mark the launch of the ground-breaking Quantidex® QPCR BCR-ABL IS Kit, manufacturer Asuragen held a series of three webinars. We covered the first webinar here, so check that out first if [...]
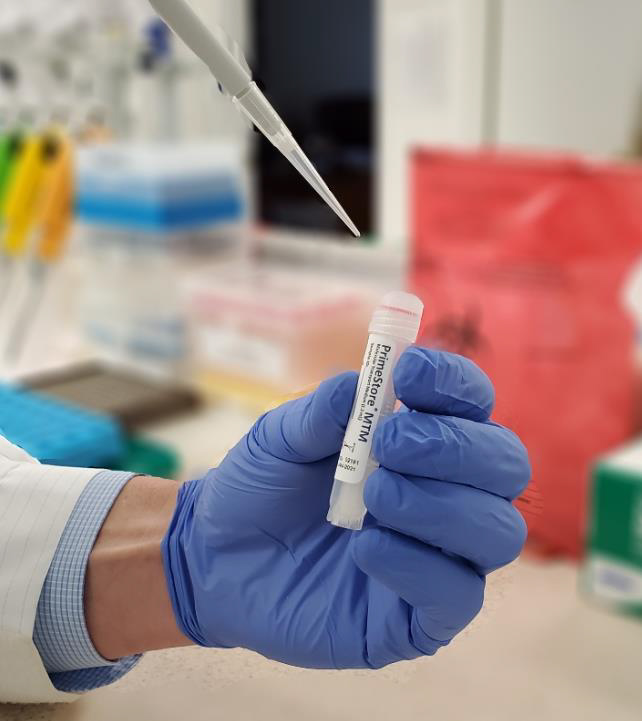
Viral Molecular Transport Medium for Safer, Faster, No Cold Chain Shipping and Testing During COVID-19
Quality molecular testing for the 2019 SARS-CoV-2 novel coronavirus requires optimised components at every stage of the testing process, beginning at sample collection. VH Bio is now able to offer a state-of-the-art [...]

Improve mental wellbeing during self isolation with the VH Bio CoLrIn BOOK
VH Bio recently designed a Histocompatibility and Immunogenetics-themed 'VH Bio CoLrIn BOOK' that was not just for fun - it was also to help promote mental wellbeing. Now, due to COVID-19 forcing [...]

VH Bio offers Asuragen Armored RNA Quant® SARS-CoV-2 Control assays in UK
VH Bio is supporting the fight against the Coronavirus pandemic with the addition of a new Asuragen-developed Armored RNA Quant® SARS-CoV-2 Control to its range, for use in developing assays in response [...]
The Evolution of CML Patient Care and the Promise of Treatment-Free Remission – Webinar
To mark the launch of the Quantidex® QPCR BCR-ABL IS Kit, manufacturer Asuragen held a series of three webinars that examine the evolving clinical landscape around CML, as well as the history [...]
Best-in-class solution for assessment of major and minor BCR-ABL fusion transcripts
The success and effectiveness of Tyrosine Kinase Inhibitors (TKIs) for treatment of Chronic Myeloid Leukaemia (CML) has altered clinical expectations as patients live longer, healthier lives. This success has, in itself, created [...]

A FLAER-based Flow Cytometry assay for the detection and monitoring of PNH clones
Paroxysmal Nocturnal Haemoglobinuria (PNH) is a stem cell disease caused by mutation of a gene involved in the synthesis of the GPI anchor of a group of surface proteins on circulating cells. [...]
NewsAlison Wortley2025-04-15T10:47:42+00:00